100% WATERPROOF
Excellent results indeed when Exposed!
Non-resorbable membranes These barriers have been widely used in guided bone regeneration (GBR) procedures in post-dental extraction sockets. In this context, one of the properties suggested for the GBR technique is that these barriers, when exposed to the oral environment, control or prevent the infiltration of connective and epithelial tissue cells, favoring the proliferation of bone cells inside the socket without bacterial infection.
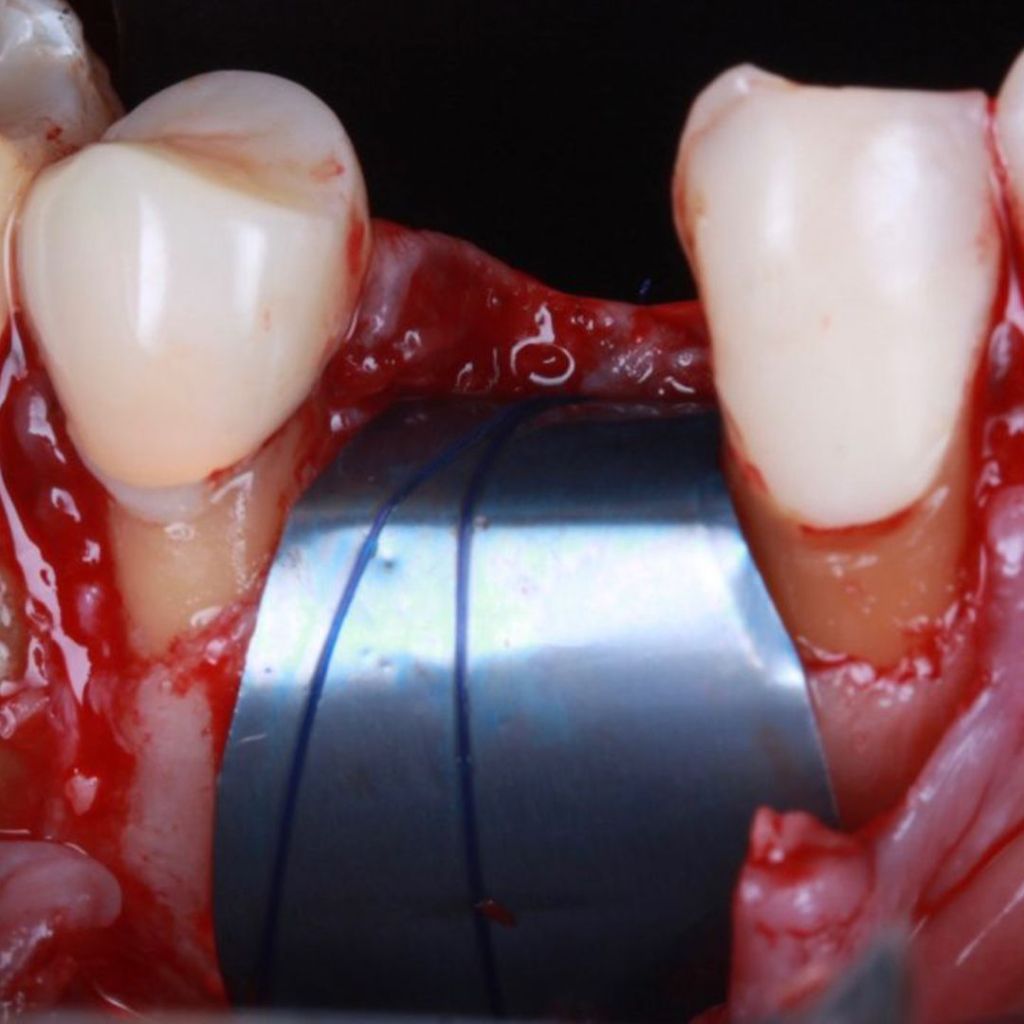
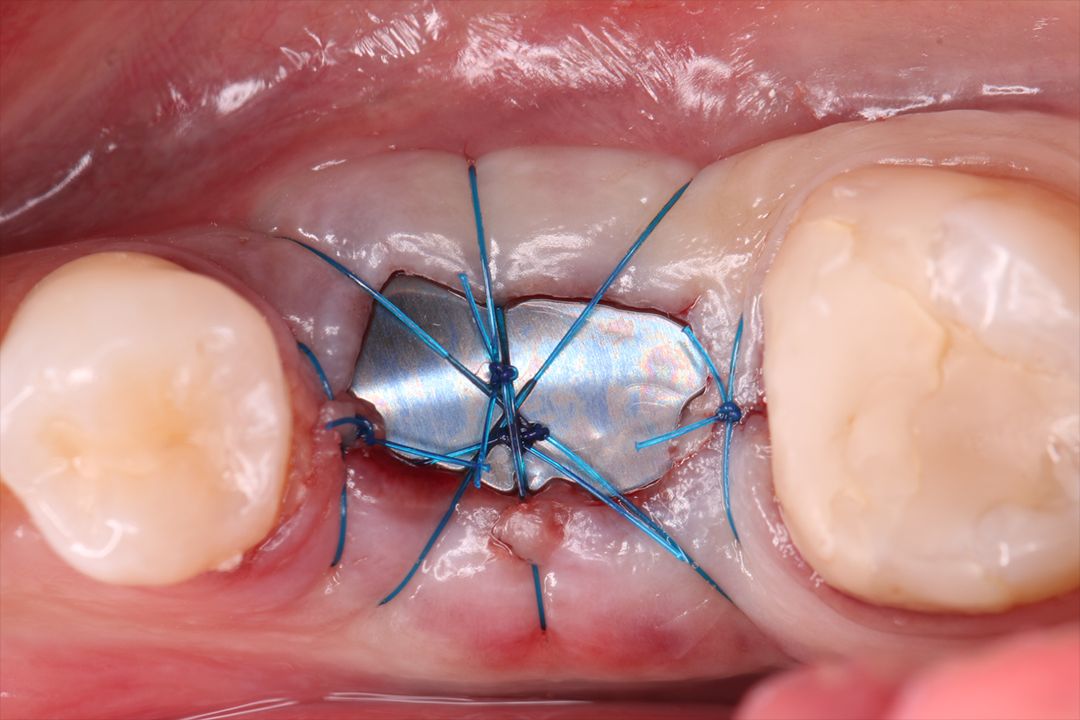
Surgitime Titanium Seal It is ideal for three-dimensional bone regeneration and was designed to be intentionally exposed during post-extraction alveolar sealing procedures, protecting the surgical wound against soft tissue invagination, which promotes resorption of the alveolar process. In this way, there is a statistically proven reduction in the rate of bone resorption. Because it is bioelectrically neutral thanks to electrochemical passivation, it contributes to uncomplicated new bone growth.
THE Surgitime Titanium Seal It is manufactured using commercially available titanium sheet. Pure Gr 1 In accordance with ASTM F67 and sterilized by Gamma Radiation.[25kGy]. The tItanium is a safe material with an excellent track record in all surgical procedures.
Topographic analysis.
The ultrastructural images revealed a rough surface, with no homogeneity in the surface structure..
Thickness ~0.04 mm
Surgical Procedures.
Recommended and designed to stay intentionally Exposed in post-extraction alveolar sealing procedures, protecting the surgical wound against invagination of soft tissues, which promotes resorption of the alveolar process. In this way, there is a statistically proven reduction in the resorption process. It should be used by shaping the mesh, taking care to completely cover the operated area with a margin that varies from 2 to 4 mm.
They should not be reprocessed, and attention must be paid to sterilization deadlines and proper instrumentation. Regarding restrictions, it is up to the professional to carefully choose the location for their implantation; that is, they must carefully consider their use in an aesthetic setting.
Due to its malleability It can be cut to fit surgical sites and, being bioelectrically neutral thanks to electrochemical passivation, contributes to uncomplicated new bone growth.
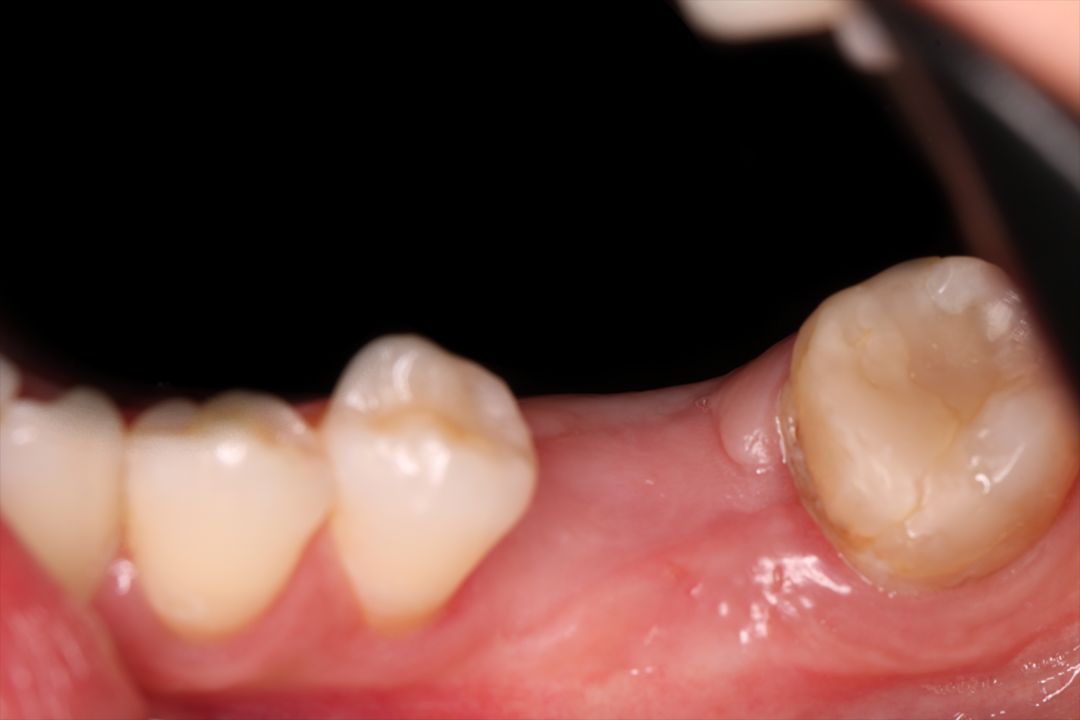
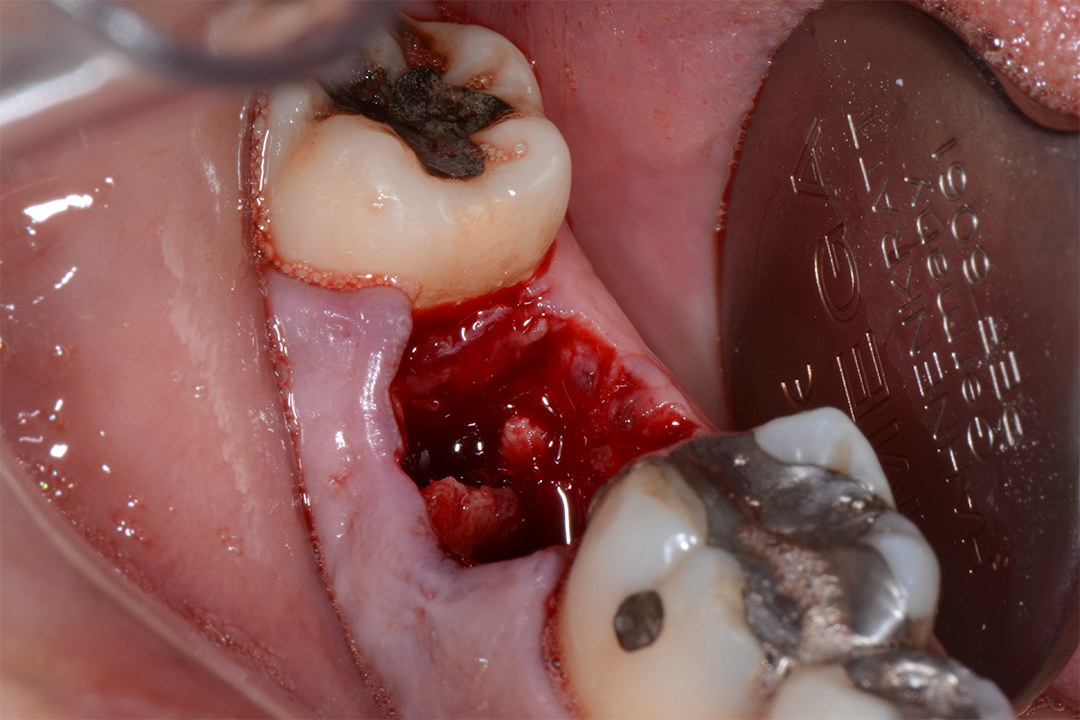
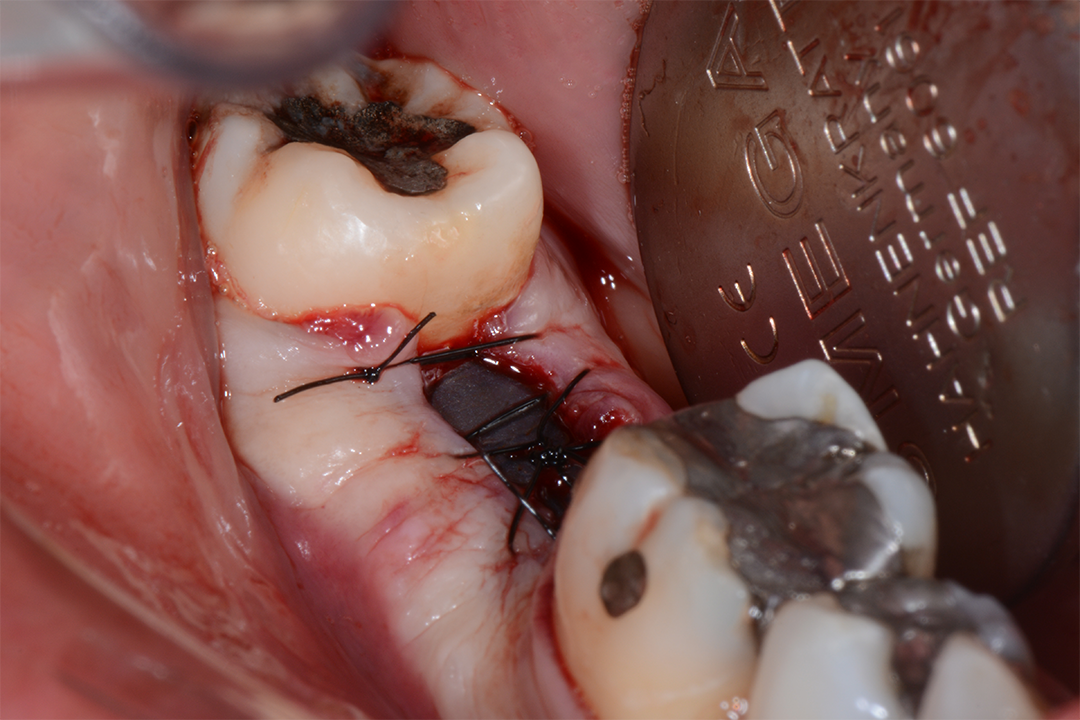
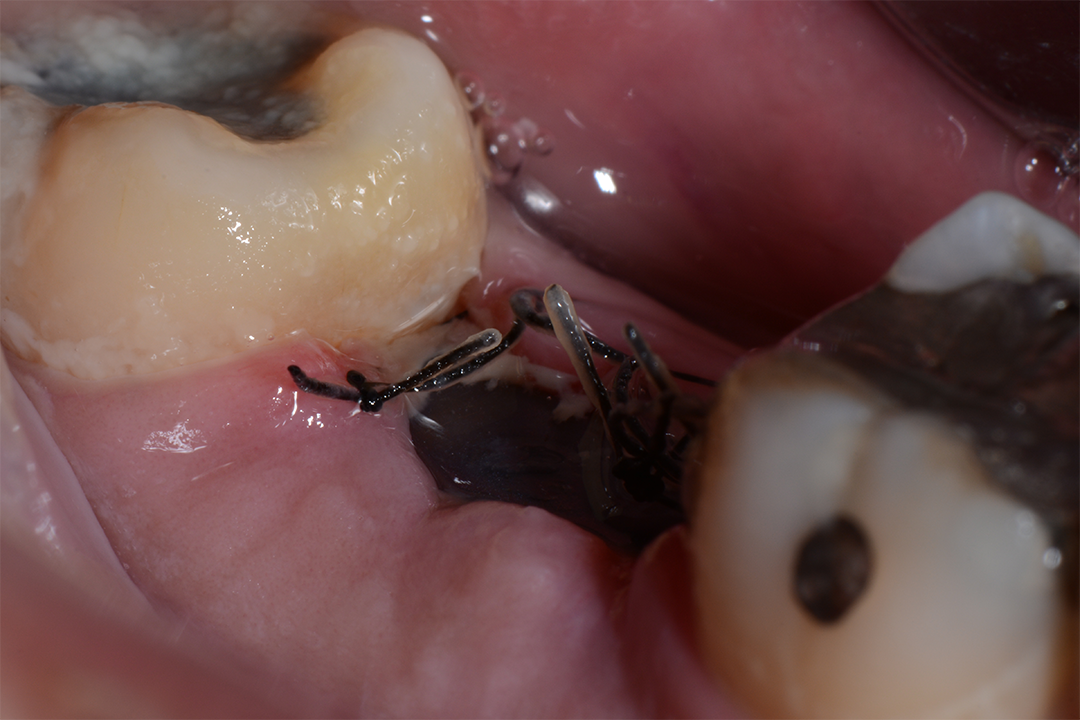
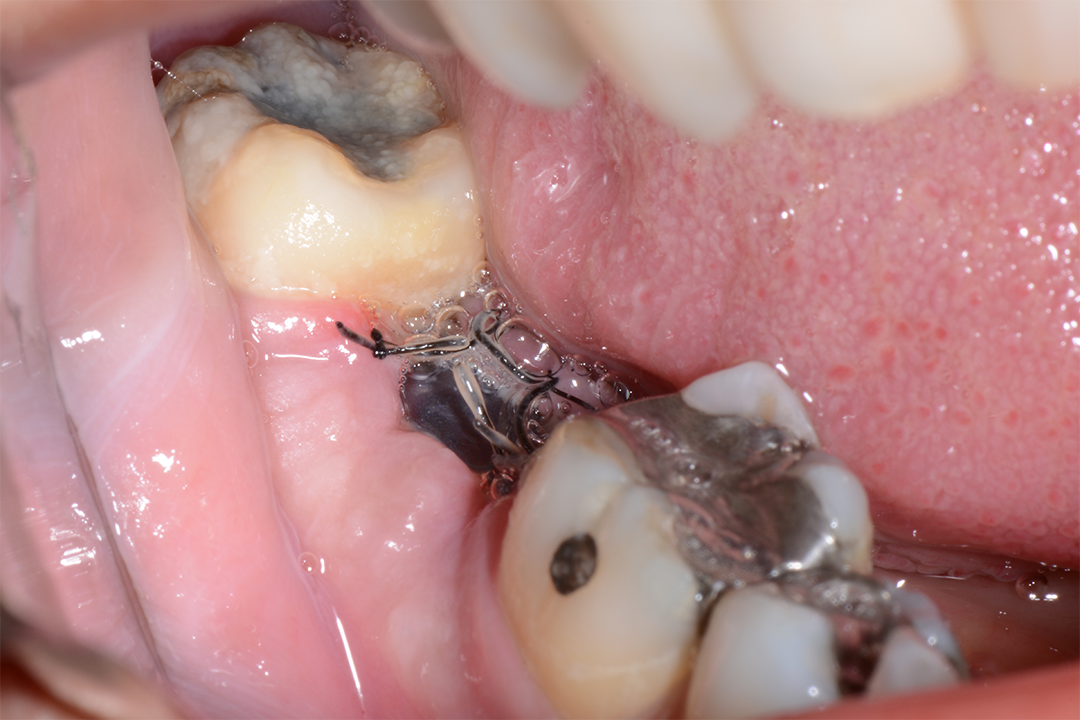
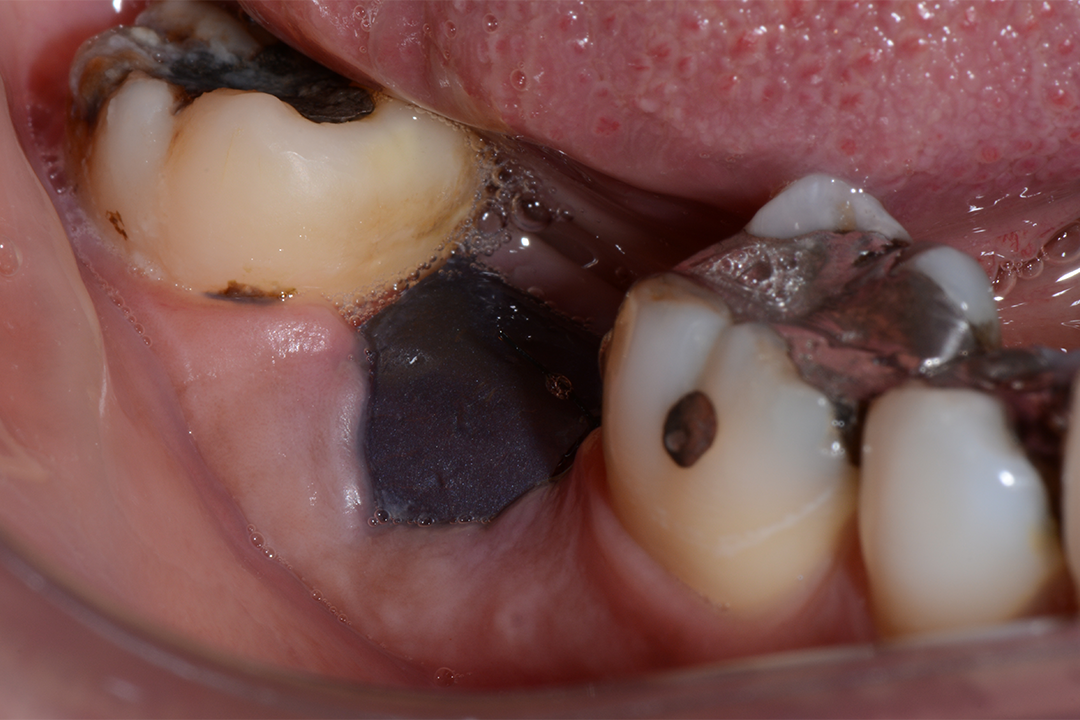
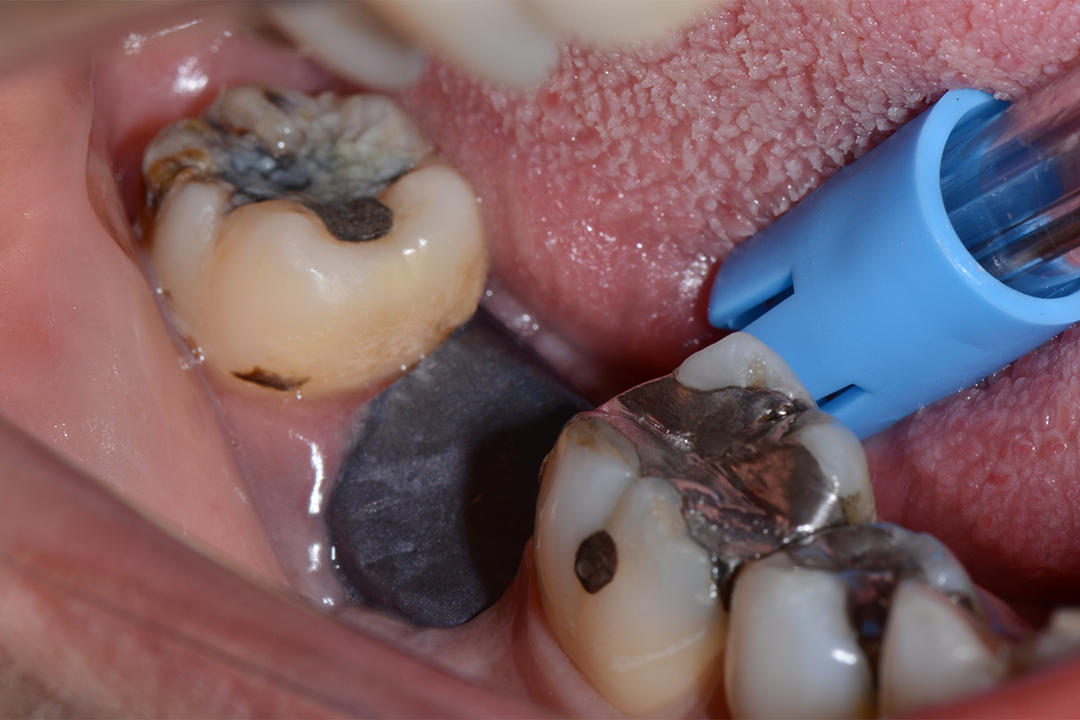
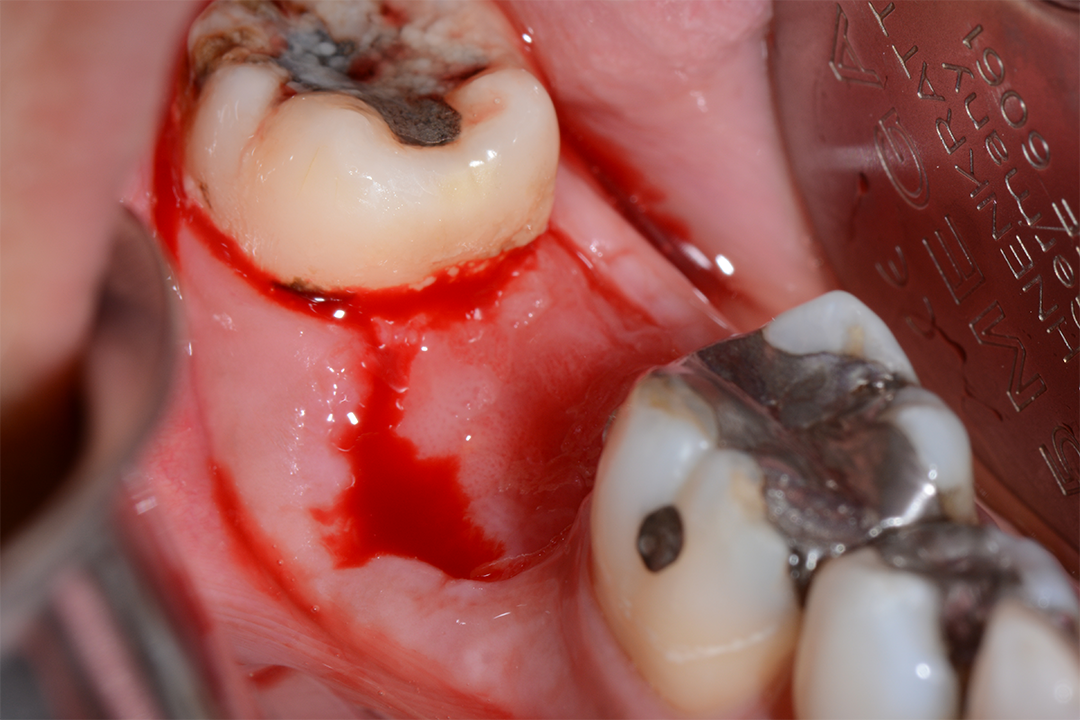
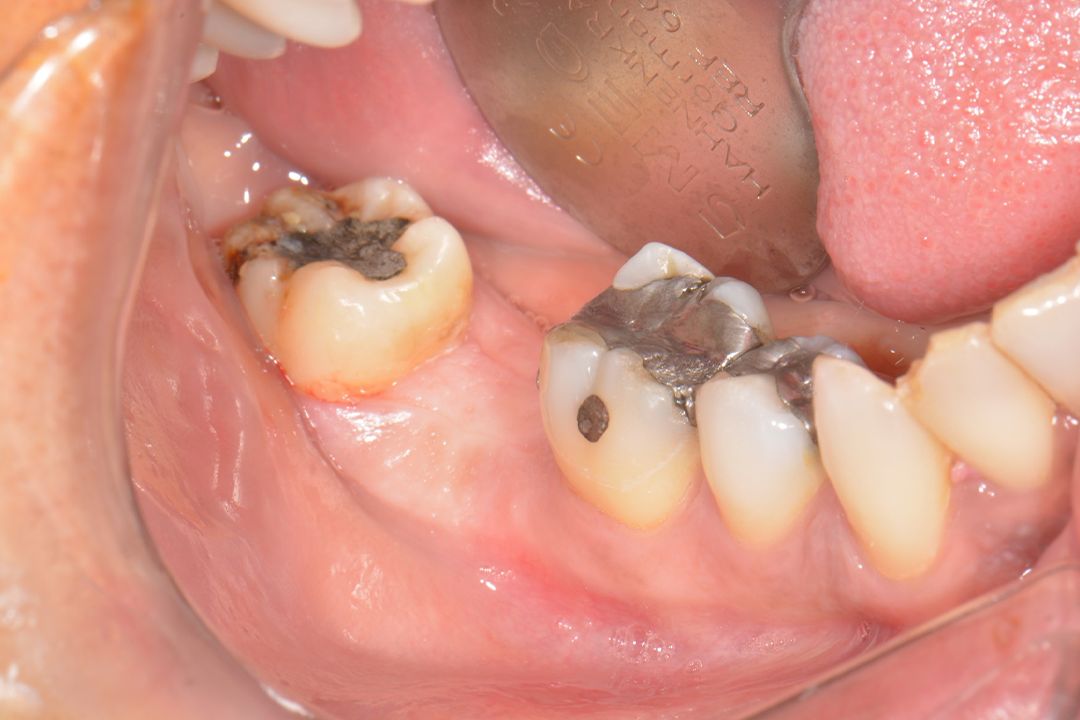
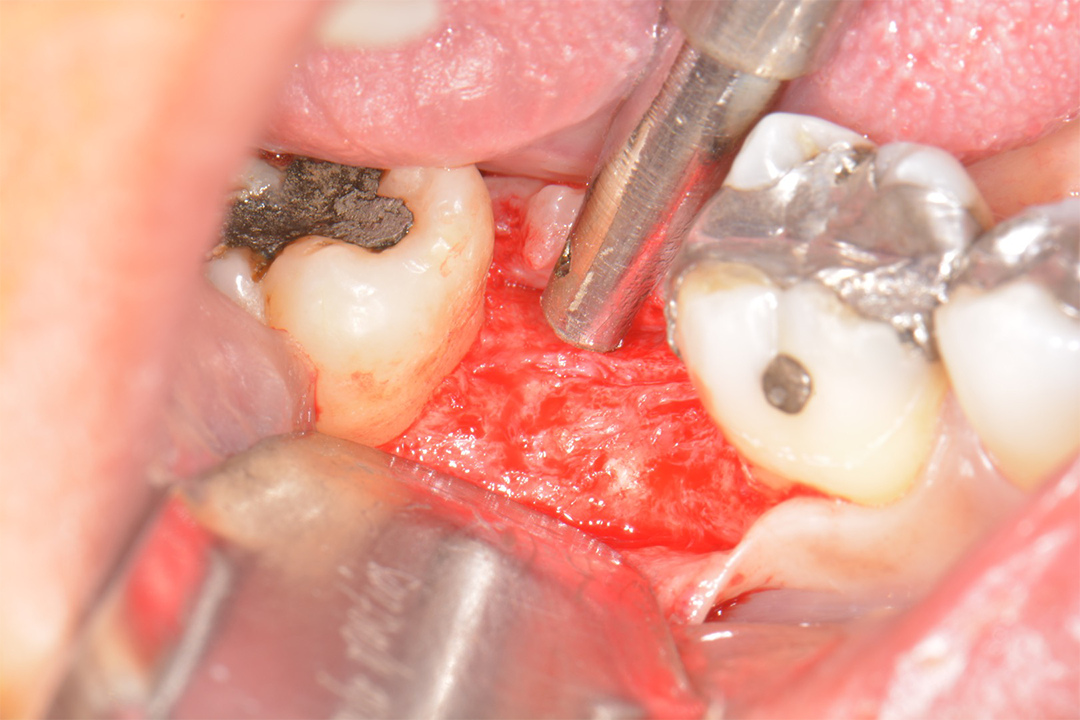
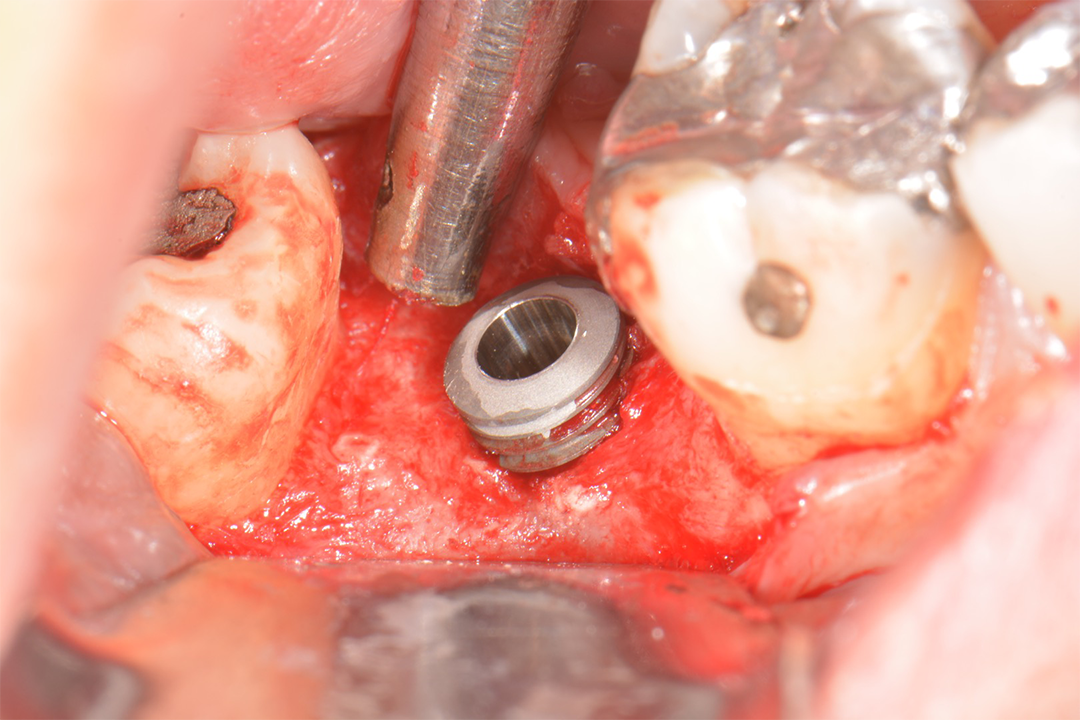
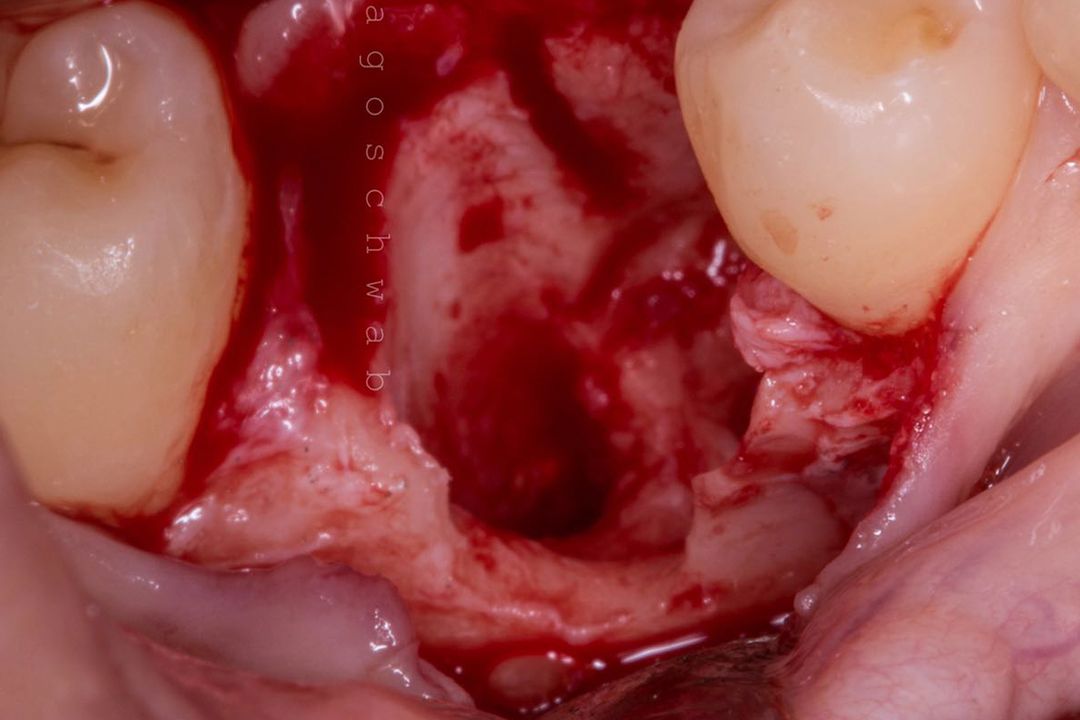
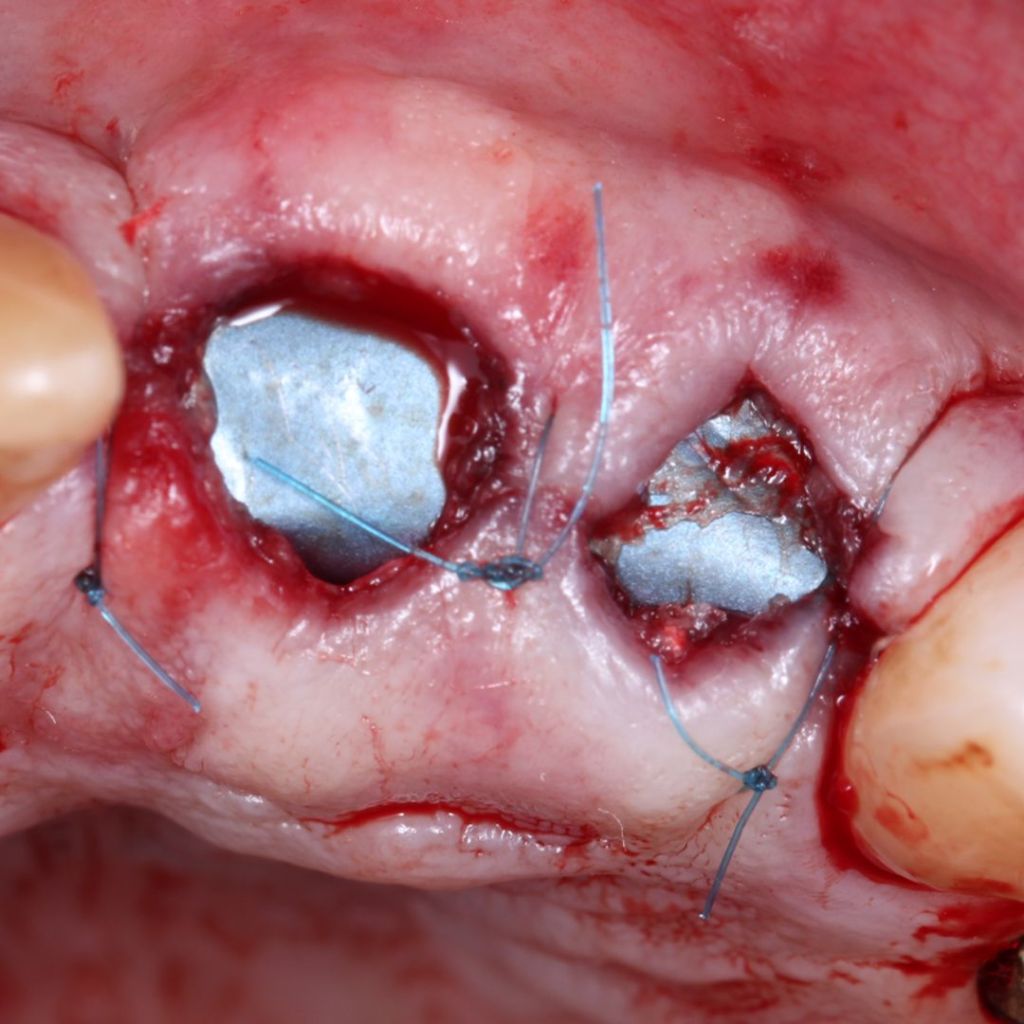
Clinical Case.
courtesy by Marcelo Faveri & Team
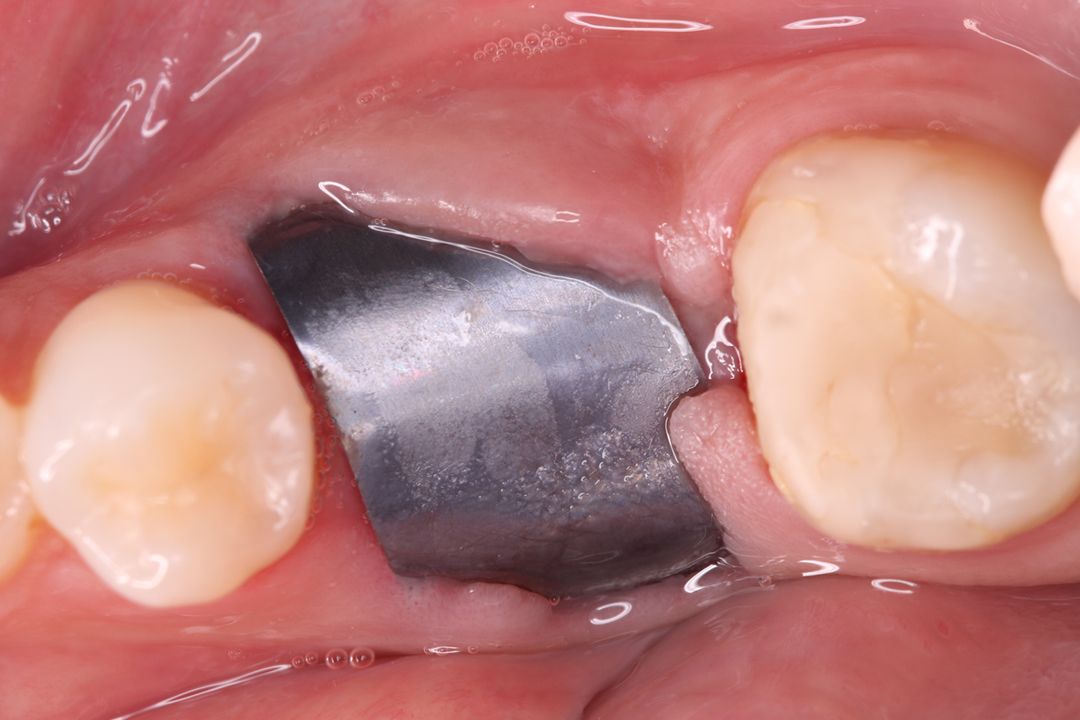
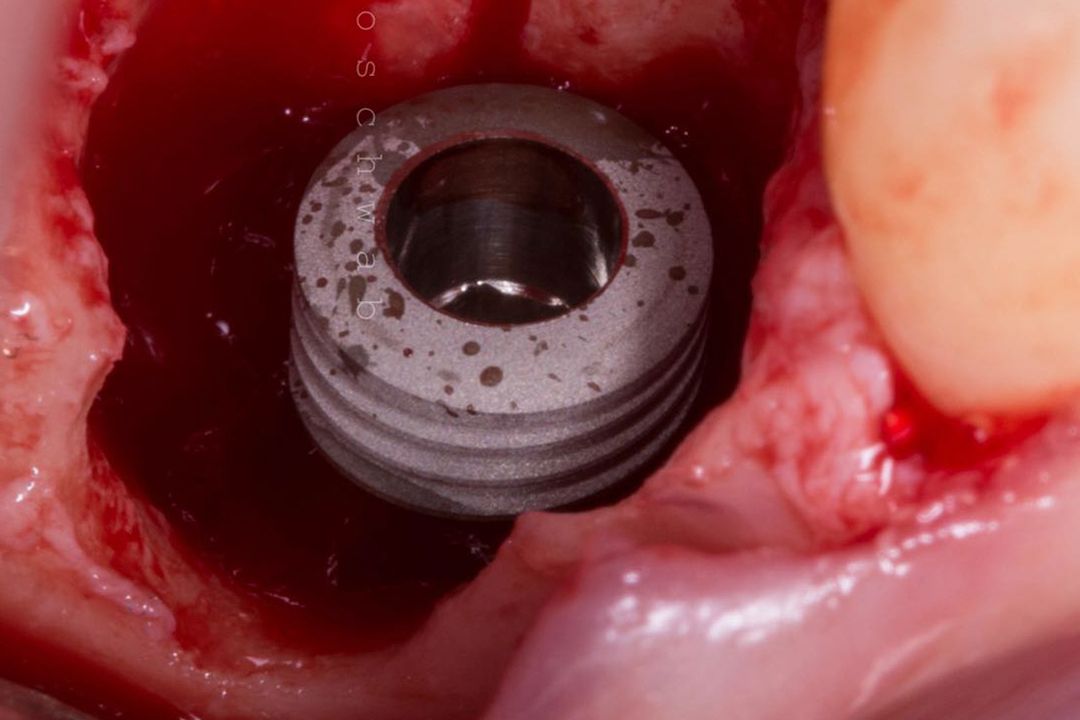
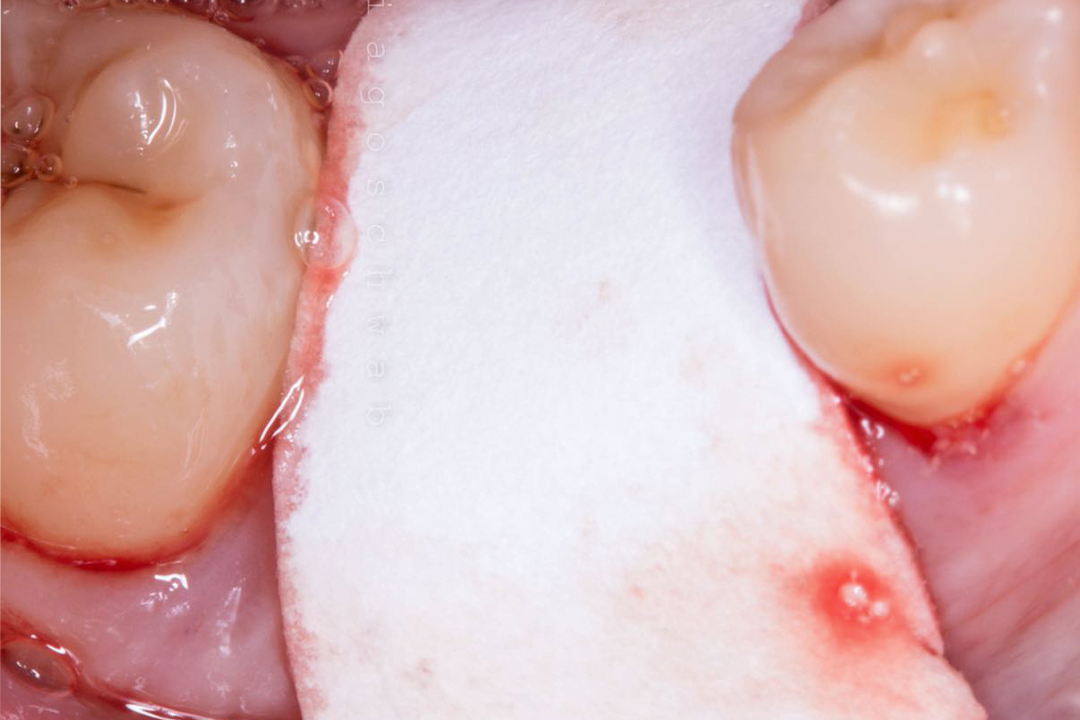
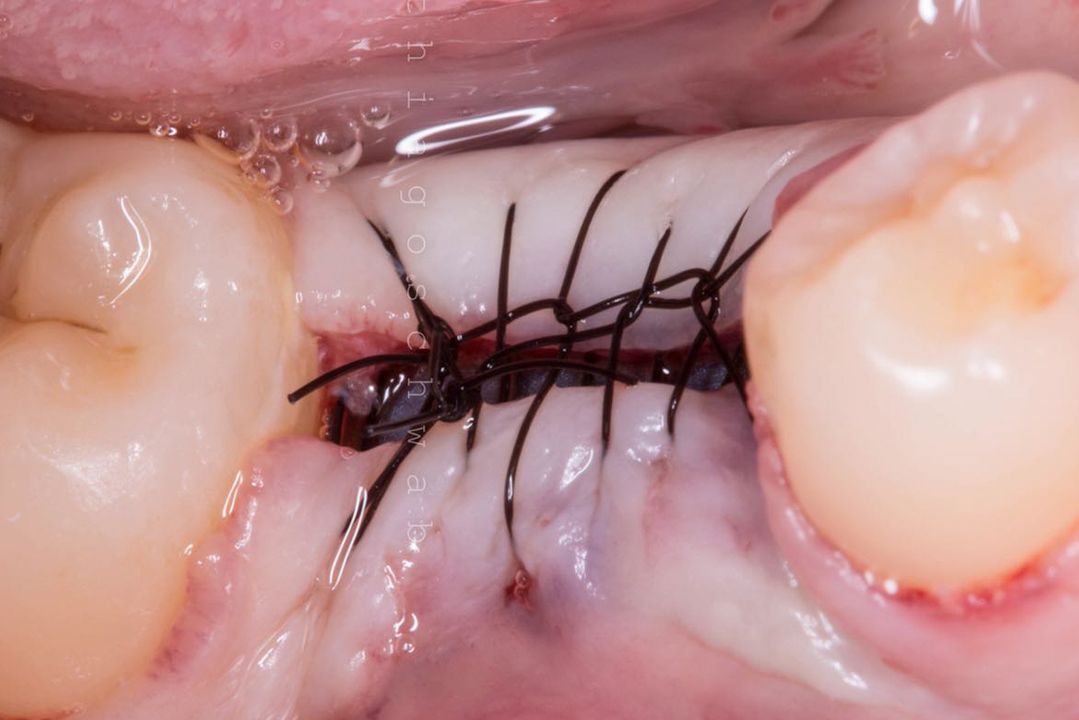
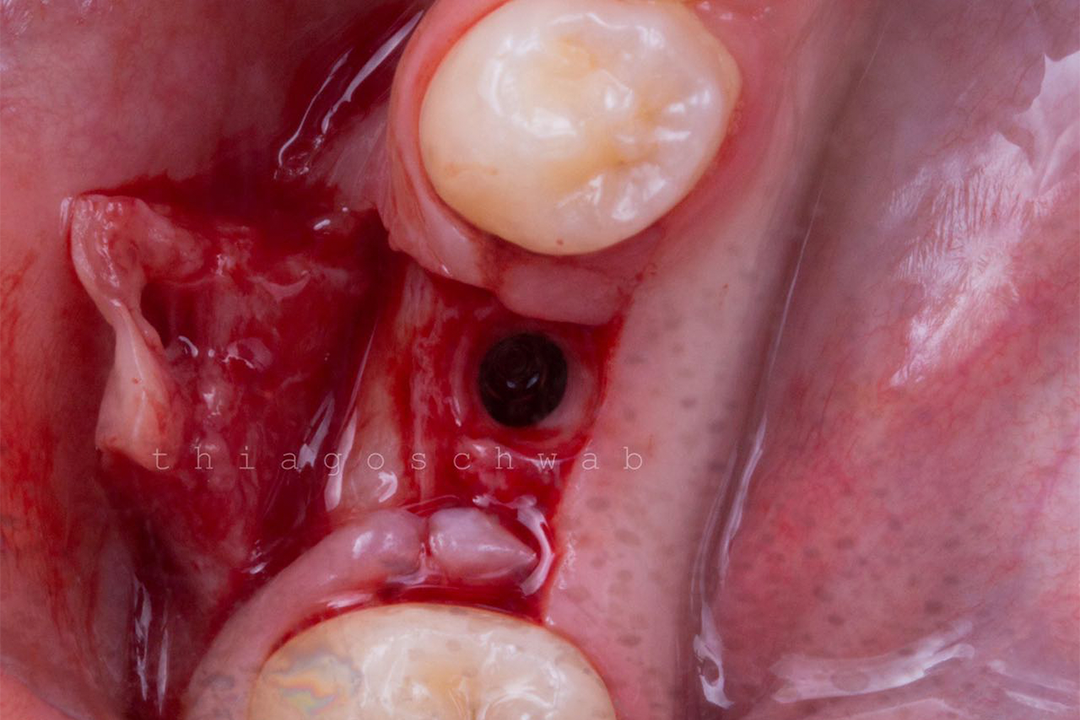
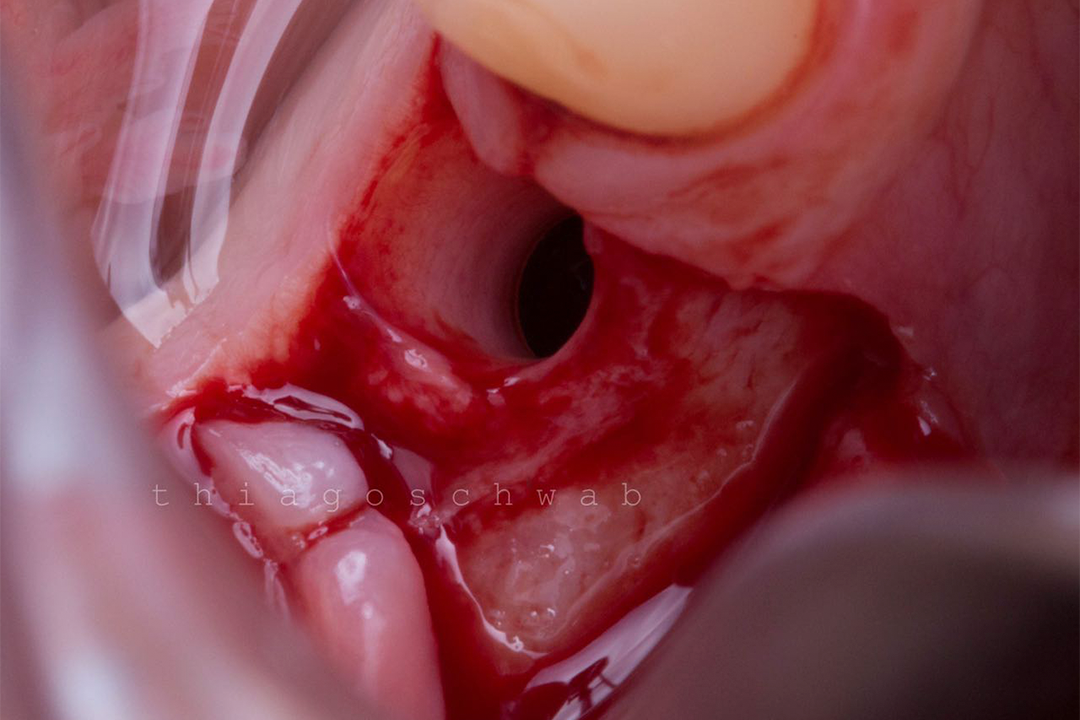
Alveolar preservation after tooth extraction for subsequent implant placement..
Use of grafting xenogeneic Bonefill® Mix with Surgitime Titanium Seal with the purpose of preservation from the edge alveolar for subsequent placement of implants in the grafted area.
THE Surgitime Titanium Seal It has excellent biocompatibilityIt’s totally… waterproof, and excludes the possibility of competition and invagination of soft tissues over bone grafts and defects.
THE Surgitime Titanium Seal It is very flexible and can be used to cover periodontal defects or alveoli and generally does not require fixation; however, if necessary, it can be fixed with Bionnovation graft screws.
The required duration for the initiation of osteoconduction is at least 21 daysand 14 days For use in cases of sealing fresh alveoli.
> Greater tissue isolation;
Self-fixation [não requer parafusos de fixação];
> Total Occlusivity;
> Easy Removal;
> Extremely low biofilm retention;
Thickness ~0.04 mm
161486 Surgitime Titanium Seal 17 x 25 mm I Thickness 0.04 mm
16890 Surgitime Titanium Seal 34 x 25 mm I Thickness 0.04 mm
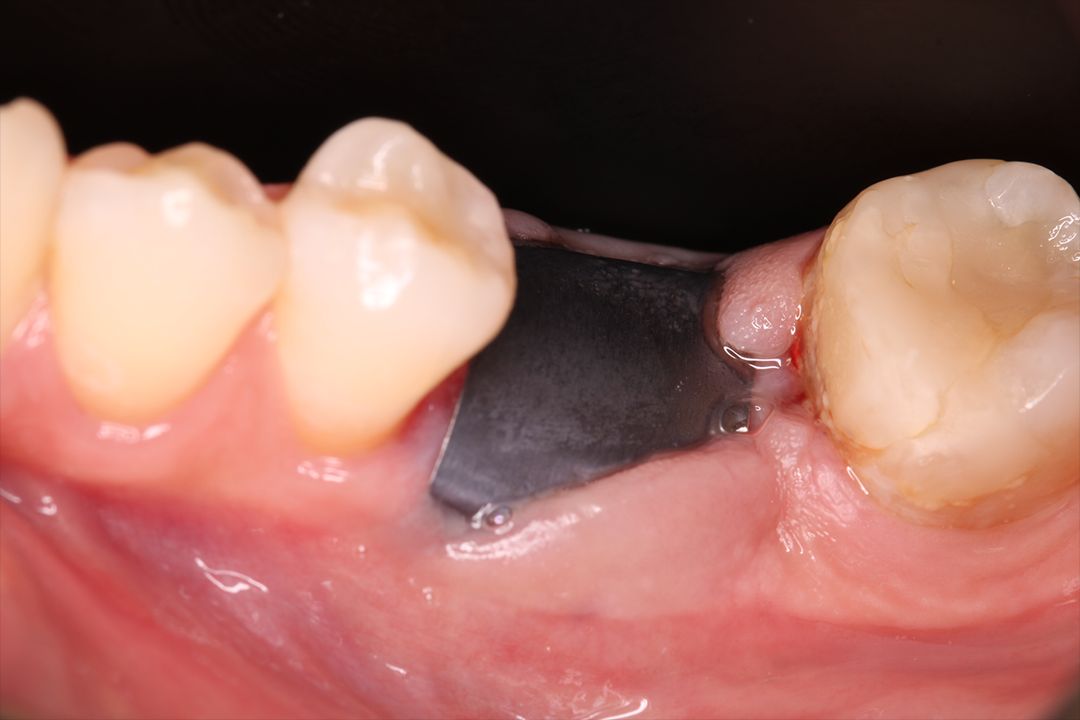
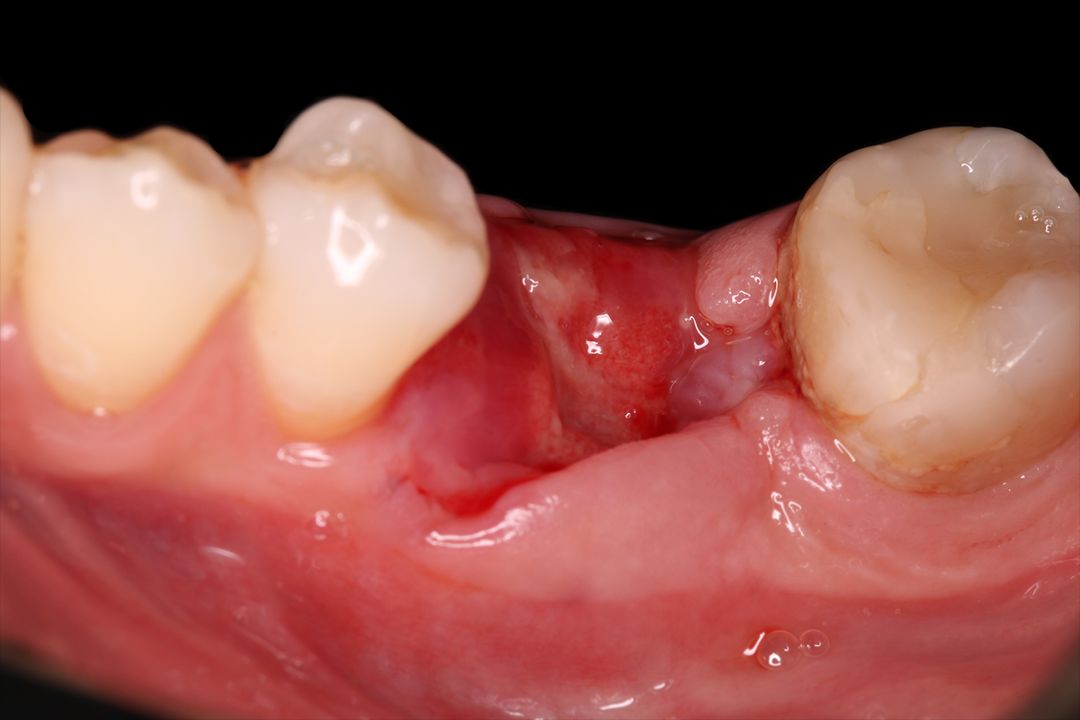
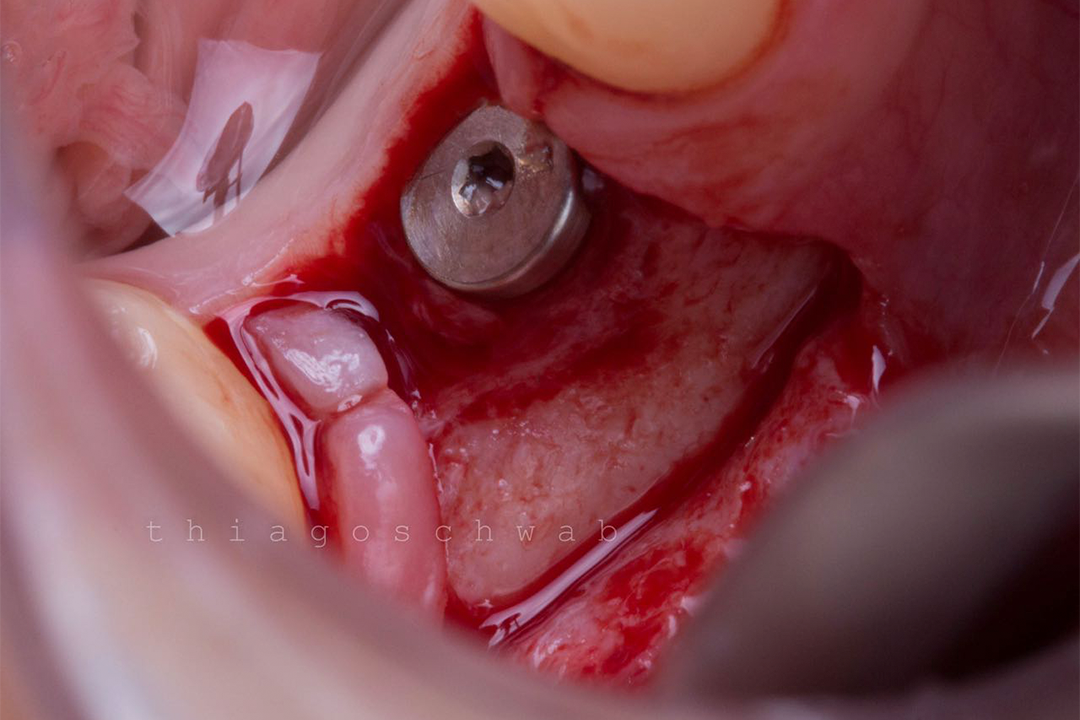
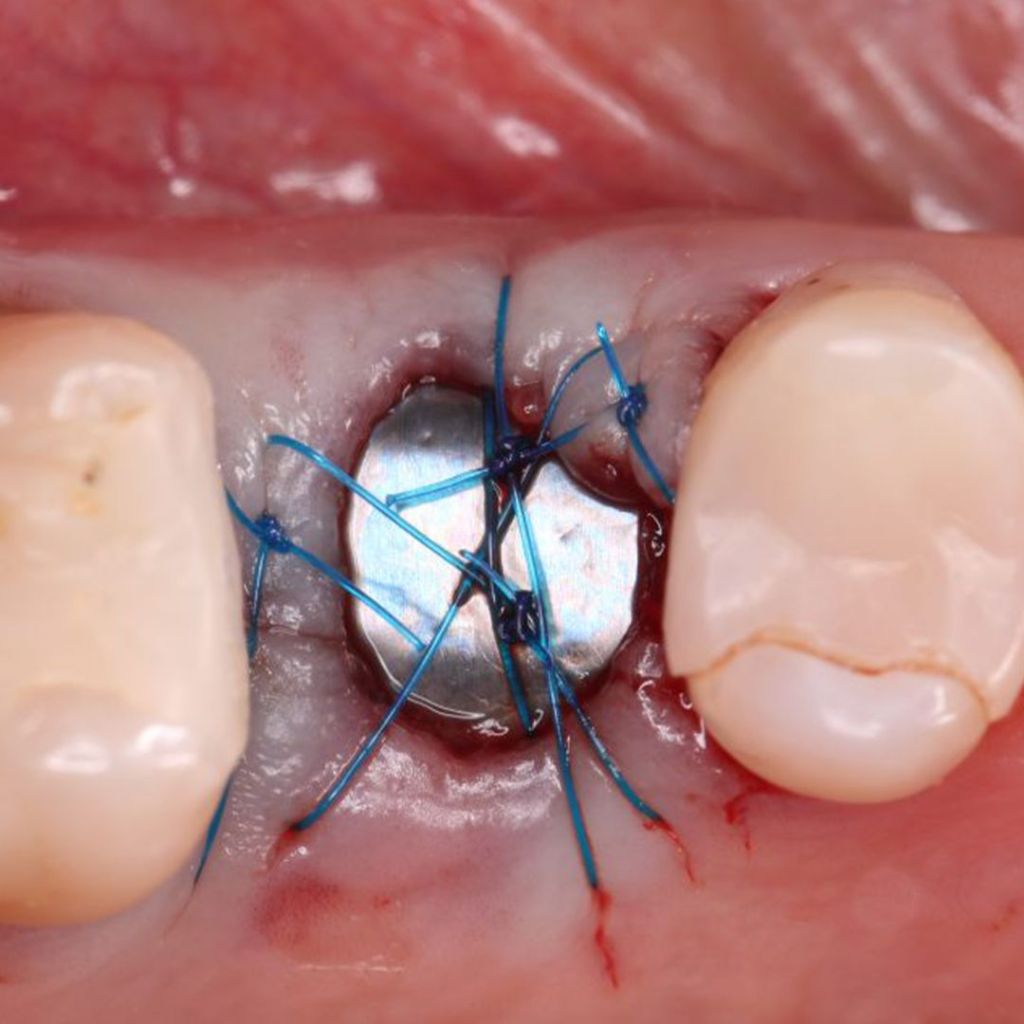
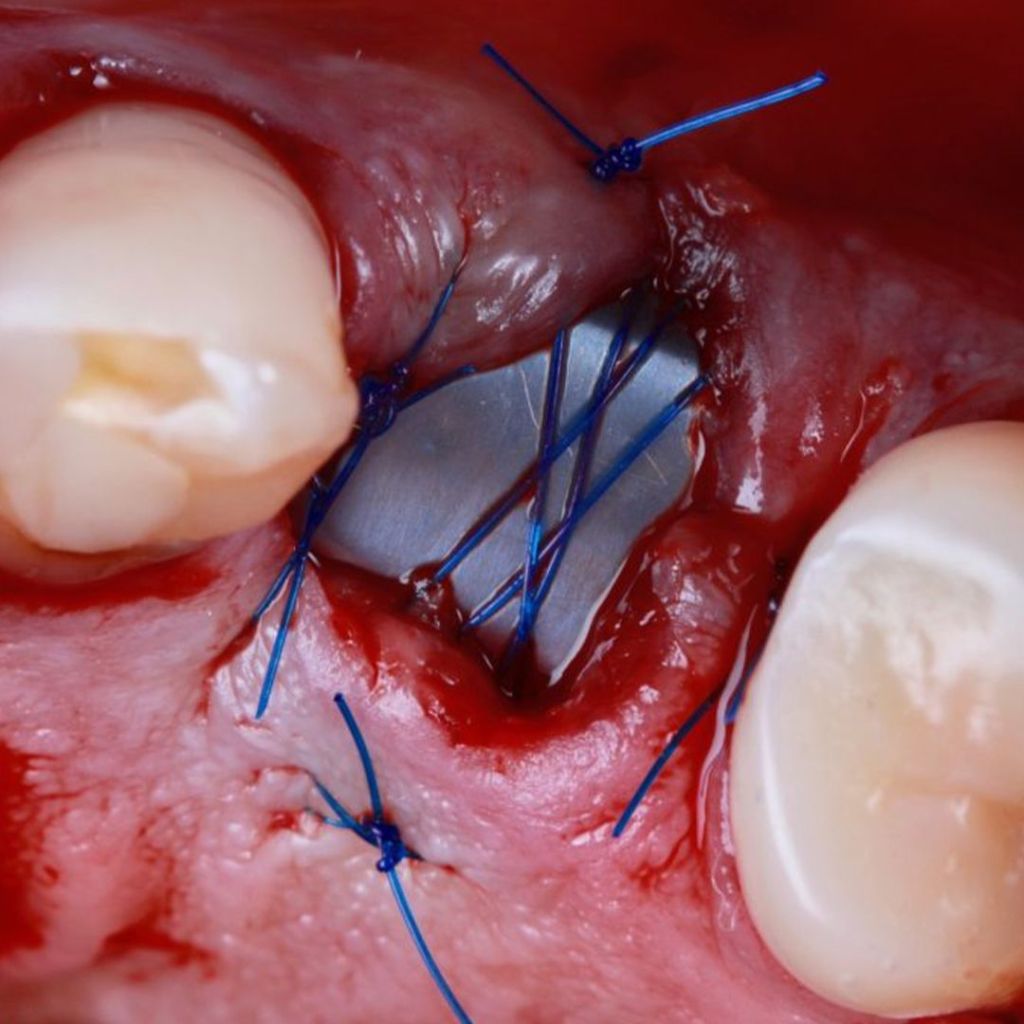
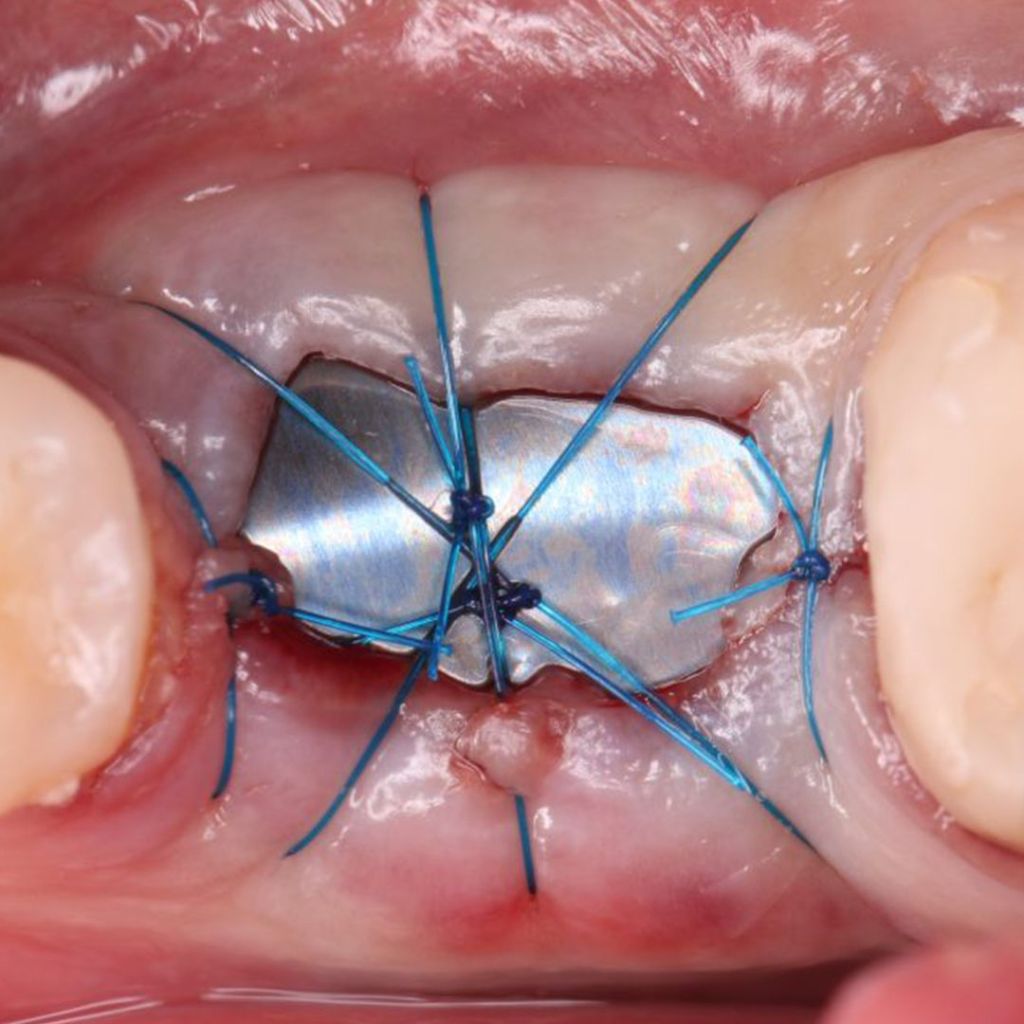
Clinical Case.
courtesy by Cassio Kampits & Team
Open healing for the preservation of the post-extraction socket.
Immediate deployment[Biomorse XPIn the posterior region with bone defects, particulate bovine mineralized matrix was used.[Bonefill® Mix#lmat_page_translation_close_translate_span# and titanium blade[Surgitime Titanium SealAfter 3 months of osseointegration, we manipulated the soft tissues with the provisional abutment on the implant. Once the tissue manipulation was complete, a transfer was performed, customizing the abutment. Case completed with a customized abutment.
Recommended reading
Marcelo Faveri, Livia Lamunier, Luciene Cristina de Figueiredo, Jonathan Meza-Mauricio, Sérgio Luís Scombatti de Souza & Bruno Bueno-Silva (2022): In vitro antimicrobial effect of titanium anodization on complex multispecies subgingival biofilm, Biofouling, DOI: 10.1080/08927014.2022.2070431.

- Who we are
- Regenerative Line
- Distributors
- Work at Bionovation
- News and Events
- Research support
- Exchange policy
- Instructions for Use (IFU)
- Contact
- Whistleblowing Channel
- Code of Ethics and Conduct
- Privacy Policy