

An excellent alternative to allogeneic and autogenous bone.
The use of bone grafts in orthopedic, cranio-maxillofacial, and dental surgeries is becoming increasingly widespread. Fresh autologous bone grafts remain the gold standard due to their properties, such as immune response and osteoinductive, osteoconductive, and osteogenic capabilities. However, their disadvantages, such as increased surgical time, small quantity obtained, and morbidity associated with this procedure, have limited their use.
Bonefill mineralized inorganic bone matrix®Bonefill has a macro and micro porous structure similar to human cortical and spongy bone and acts as an osteoconductive mechanism, promoting bone growth and regeneration. Over time, Bonefill® It is partially remodeled by the action of osteoclasts and osteoblasts, making it a viable alternative to autologous bone in defects suitable for its use and indication..
Developed for bone regeneration in reconstructive surgeries, the Bonefill® It is a natural bone substitute obtained from cortical and spongy bone of the condyles. femur bovine. The highly purified osteoconductive mineral structure is produced through a multi-phase process, complying with the safety regulations stipulated by control agencies. Fresh bone is crushed and undergoes a series of baths that solubilize all organic structures, leaving only its mineral portion and thus preventing the induction of possible immunogenic processes in the body. Products made from mineralized bovine bone matrix are expected to be incorporated within a minimum of 6 months, depending on the regenerative potential of the treated defect.
The Bonefill® in the form of granulesIt is available in the following models: Bonefill® Dense, Bonefill® Mix and Bonefill® Porous, and in the form structural It is available in the following models: Bonefill® Cortical Lamina, Bonefill® Porous Block, Bonefill® Porous Cylindrical and Bonefill® Porous pre-molded in a special shape.
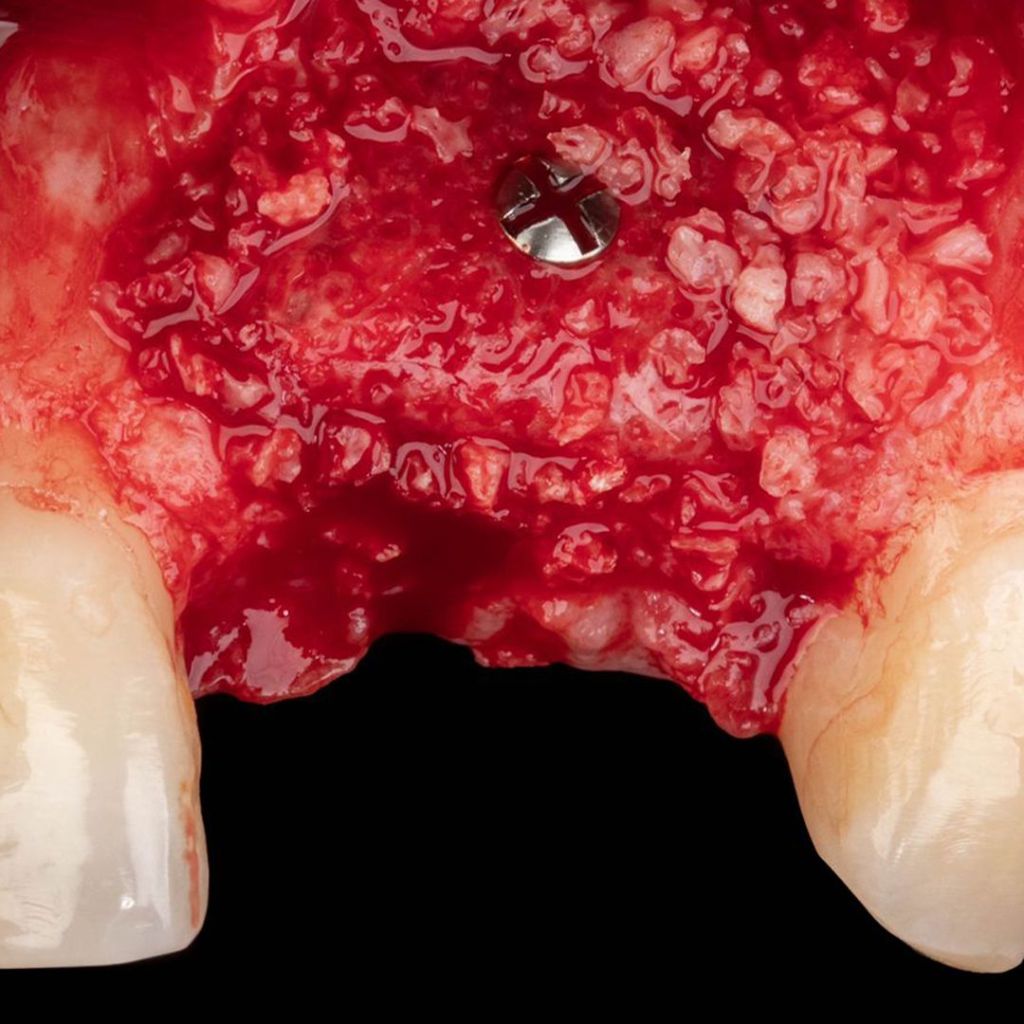
Topographic analysis.
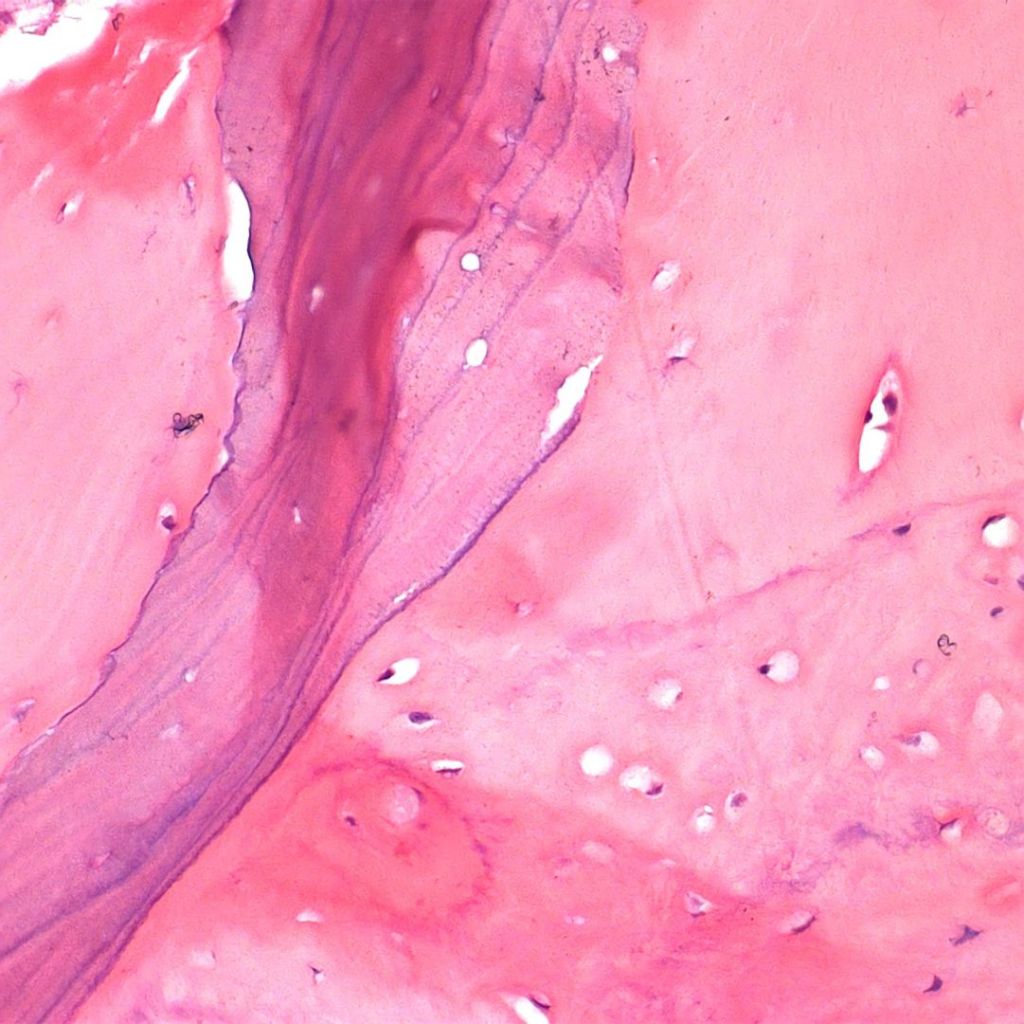
The microscopic structure of bone grafts Bonefill®Analysis by SEM/EDS revealed that the product exhibits a typical structure of human cortical and cancellous bone, with interconnected pores, and that, after the physical-chemical process to which the raw material is subjected for manufacturing, it maintains the characteristic structure of this type of tissue. The presence of pores and the maintenance of the trabecular crystalline structure of the bone are fundamental factors for the success of the cellular phenomena of osteoprogenitor cell deposition on the graft, its resorption, and the formation of new bone in its place.
Histological analysis.
With a calcium phosphate ratio that mimics the composition of human bone and a low crystalline structure, the Bonefill® It is accepted by the human body as a suitable structure for bone formation.
Bonefill characteristics® They allow for rapid hydration and consequently blood absorption, which is key to initiating the regenerative process in bone defects. Furthermore, in addition to allowing the accumulation of growth factors from the blood, they also provide an excellent three-dimensional framework, offering a suitable and favorable environment for bone regeneration and reconstruction.
The Bonefill combination® Using autogenous bone adds biological activity (osteoinductive and osteogenic properties of autologous bone), promoting faster regeneration and improving the formation of new bone.
The appropriate healing period depends on the patient and the initial type of defect, and should be decided by the professional based on an assessment of each individual case. In current studies, we recommend a minimum healing period of six months before re-entry to ensure the stability of particle integration.
Clinical investigations have confirmed the mechanism of action of Bonefill.®They very easily absorb large amounts of blood, thus initiating microcoagulation within the graft itself and therefore greatly improving graft integration; the first few weeks are then necessary for cellular colonization of the graft, however, this time frame is also necessary for the degradation of the thin polymeric film, which progressively fades, leaving the mineral structure for the cells to consolidate and promoting the formation of new living bone (also through the formation of new vessels); subsequent months are necessary for the integration of the graft with the patient’s native bone, also due to vascularization and bone neoformation within the graft via complete remodeling.



- Who we are
- Regenerative Line
- Distributors
- Work at Bionovation
- News and Events
- Research support
- Exchange policy
- Instructions for Use (IFU)
- Contact
- Whistleblowing Channel
- Code of Ethics and Conduct
- Privacy Policy








