
Bovine collagen membrane
with exceptional handling properties.
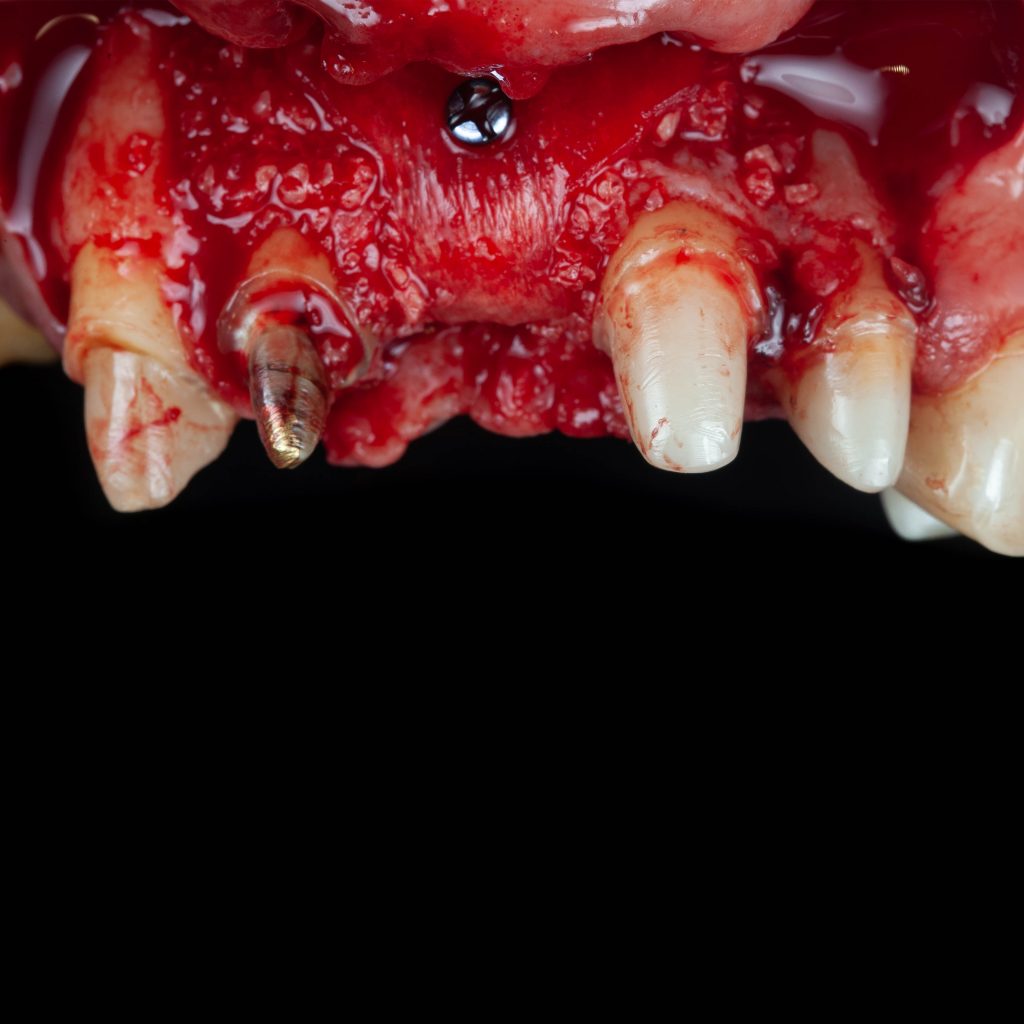
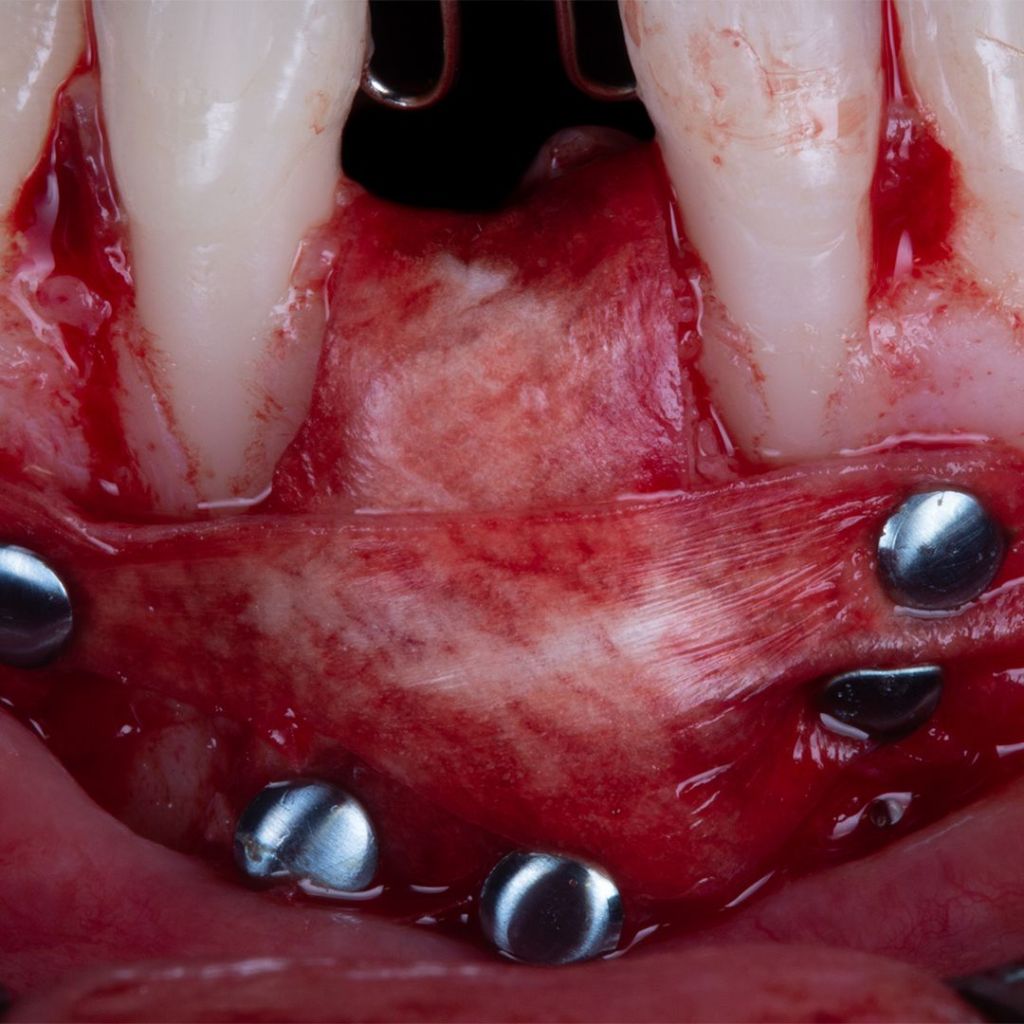
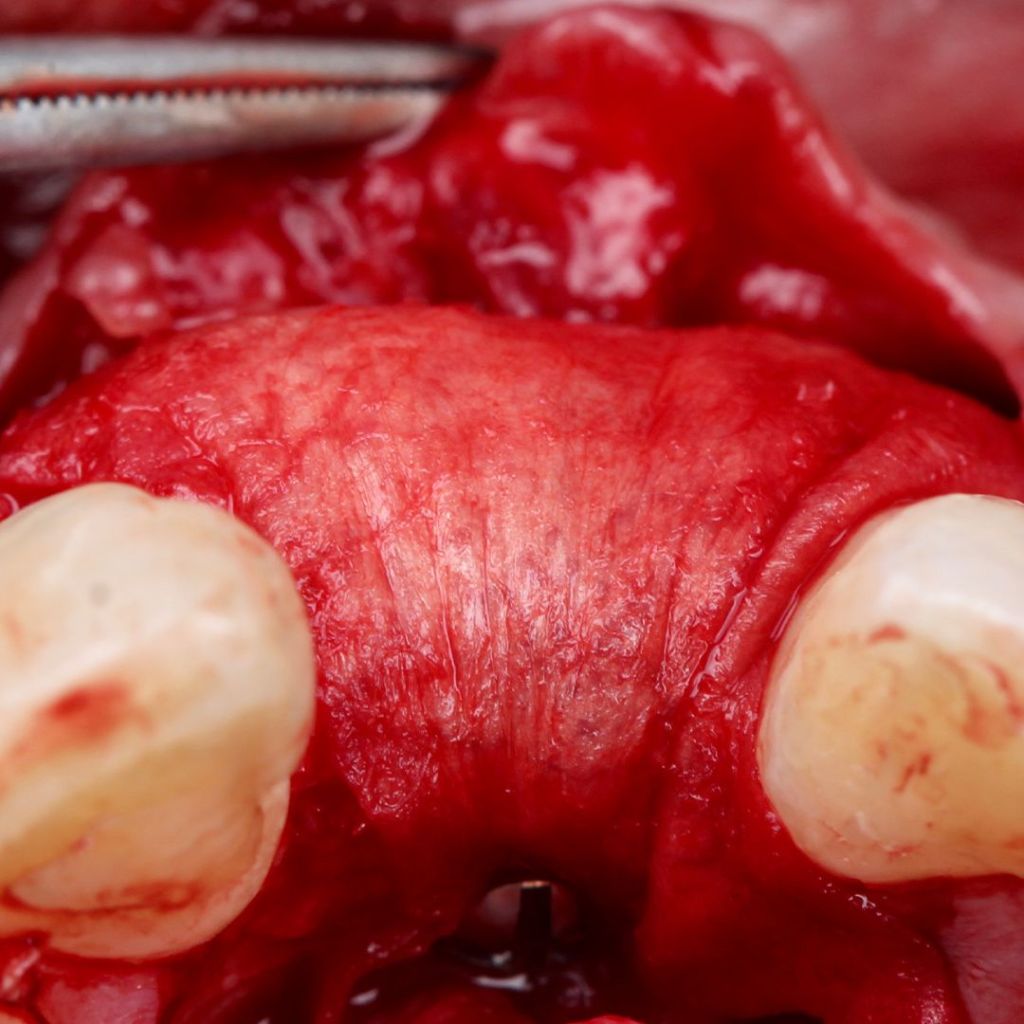
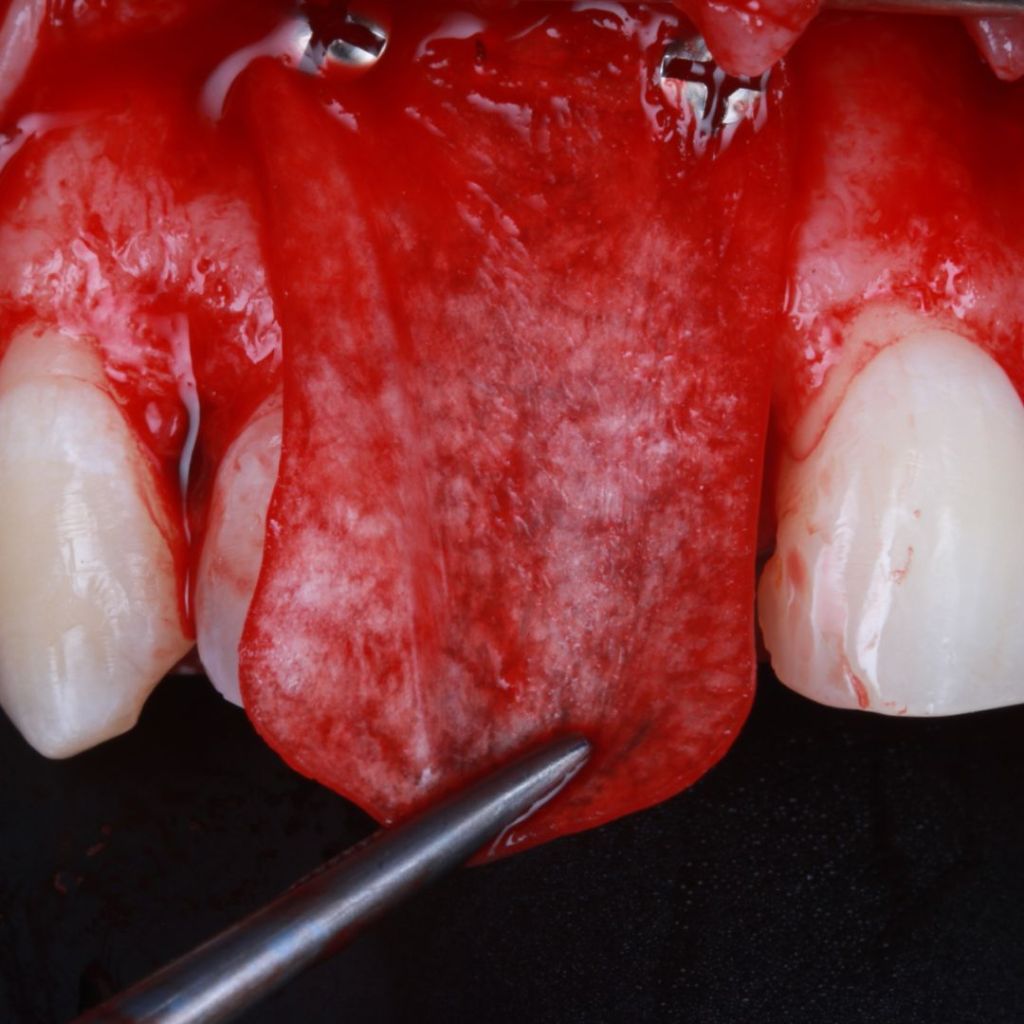
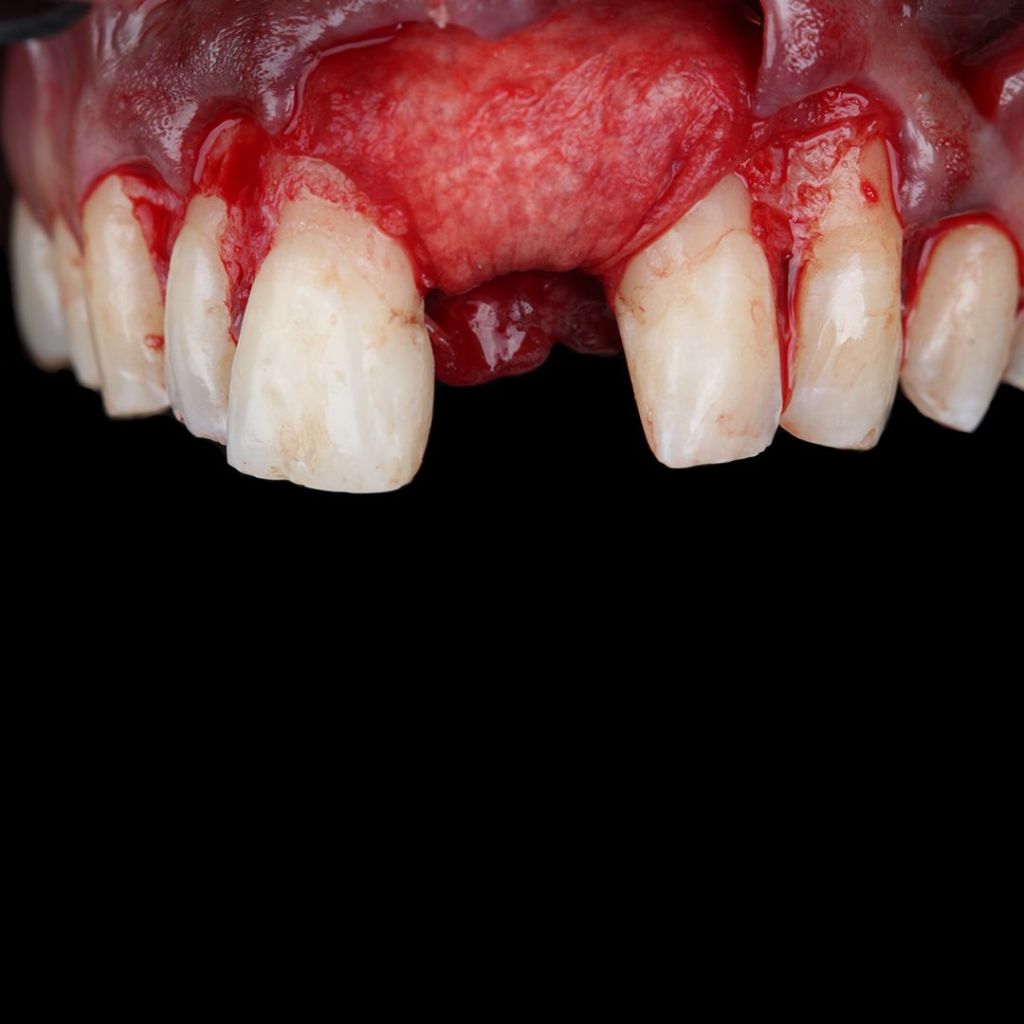
Surgitime Collagen It is an implantable and resorbable acellular matrix of bovine pericardium, designed for use as a barrier in bone regenerative procedures in implantology, periodontics, and maxillofacial surgery..
Obtained through acellularization mechanisms from bovine pericardium, the final product is a highly pure natural collagen membrane that acts as a regenerative biological barrier when implanted over bone grafts and beneath gingival tissues, in so-called guided bone regeneration procedures..
In these procedures, barriers or membranes serve to physically separate tissues, preventing competition between soft tissues for space and nutrition from the bone graft. In other words, they prevent the unwanted migration of epithelial and connective tissues into the bone defect filled with the graft during bone remodeling and formation.
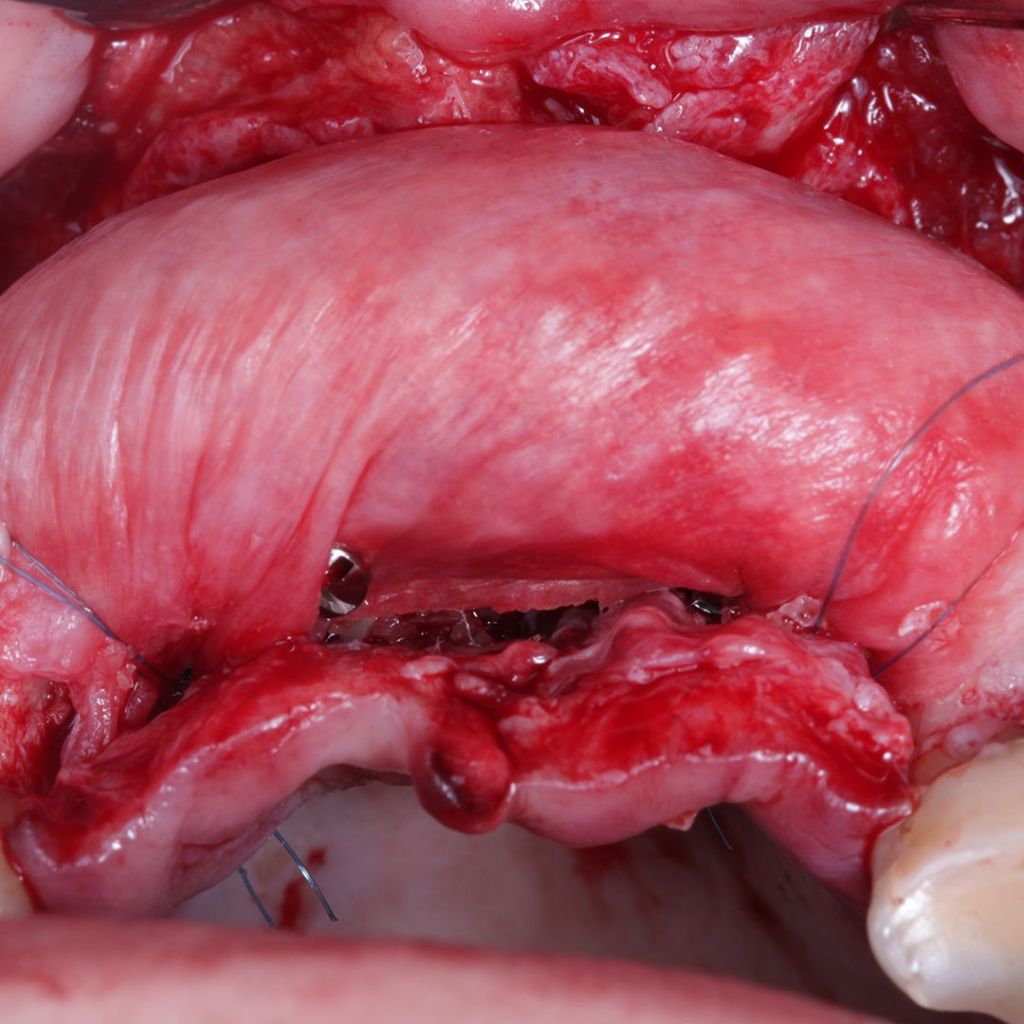
Mechanical strength
Regarding the mechanical resistance of the bovine pericardium, the Surgitime Collagen It possesses biomechanical properties that guarantee high resistance to tension and rupture in various directions, and can physically contain bone graft materials, promoting an immobile environment conducive to bone graft integration. It allows for excellent fixation using graft screws, tent screws, tacks, or sutures..
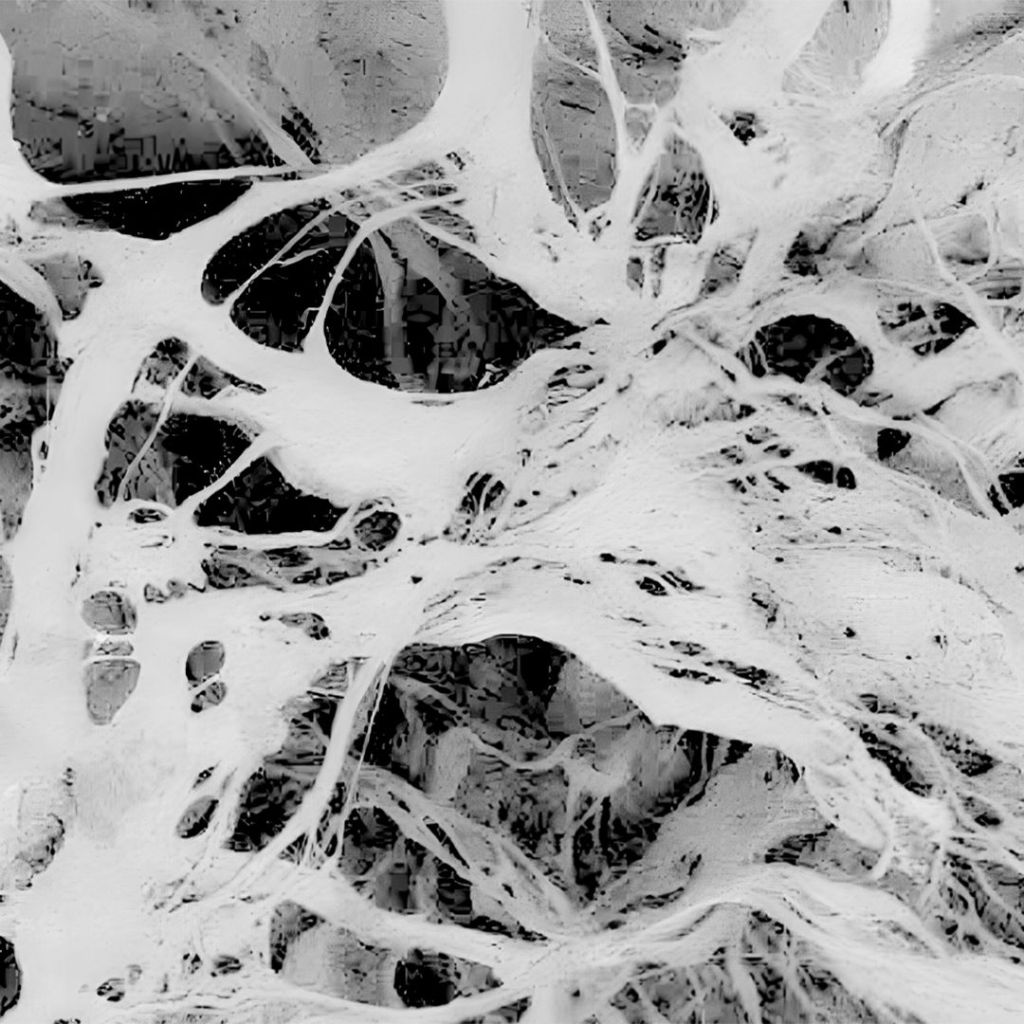
Topographic Analysis
THE Surgitime Collagen It has a porous structure; the size of its pores is small enough to occlude gingival cells but large enough to allow vascularization, proliferation, and cell maturation, essential conditions for the natural processes of bone and connective tissue remodeling and reconstruction. We can observe, from images obtained through scanning electron microscopy (SEM), a homogeneous surface with a high degree of similarity to human collagen..
Recommended for use as a barrier in bone regenerative procedures in implantology, periodontics, and oral and maxillofacial surgery. Alveolar ridge reconstructions for prosthetic rehabilitation treatment, alveolar ridge augmentation prior to or simultaneous with implantation, ridge augmentation or volumetric preservation in immediate implant procedures, filling of bone defects after root resection, cyst removal surgery or removal of impacted teeth, guided regeneration procedures in periodontal defects, access closure in bone grafting surgery in maxillary sinuses.
CompositionPurified natural collagen Type I
ConformabilityAdaptable to the location of the defect.
Structural integrityNon-brittle
PorosityAverage pore size 19.52μm
Small enough to occlude the gingival cells but large enough to allow the passage of fluids, nutrients, and plasma proteins, which are necessary to aid healing.
Reticulated: No
SterilizationGamma radiation
Lifespan3 years
Excellent soft tissue healing..
One of the main advantages of the natural porous structure of Surgitime Collagen Pericardium is its excellent healing characteristic resulting from its good vascularization and tissue integration. Even in cases of dehiscence, the soft tissue usually heals well and no additional growth is necessary.
Easy to cut [seca ou hidratada]It does not adhere to instruments. Both sides can come into contact with the defect. It can be used in conjunction with synthetic or natural bone grafts in all clinical situations requiring containment of the filling material and tissue isolation.
Safe.
The production process of Surgitime Collagen It is a standardized and controlled process that subjects bovine-derived tissue to a multi-step sequence of reagents that acellularizes it, while preserving the characteristics of type I collagen, resulting in a high degree of purity, biocompatibility, and freedom from heavy metals and any biological contamination.
A series of tests of biocompatibility In vitro and in vivo studies were conducted to evaluate the safety of Surgitime Collagen as an implantable material. The device has passed all applicable tests for the ISO 10993-1 biological evaluation of medical devices.
Biocompatible, free of heavy metals and any biological contamination. It is acellular, non-cytotoxic, non-immunogenic and non-pyrogenic.
Step by Step.
161272 Surgitime Collagen Pericardium 15 x 20 mm
161273 Surgitime Collagen Pericardium 30 x 20 mm
161276 Surgitime Collagen Pericardium 40 x 30 mm
- Who we are
- Regenerative Line
- Distributors
- Work at Bionovation
- News and Events
- Research support
- Exchange policy
- Instructions for Use (IFU)
- Contact
- Whistleblowing Channel
- Code of Ethics and Conduct
- Privacy Policy
